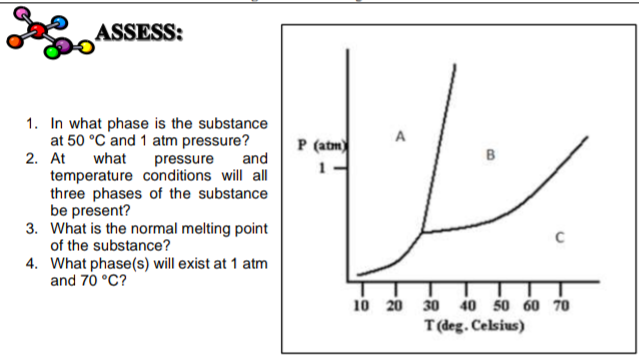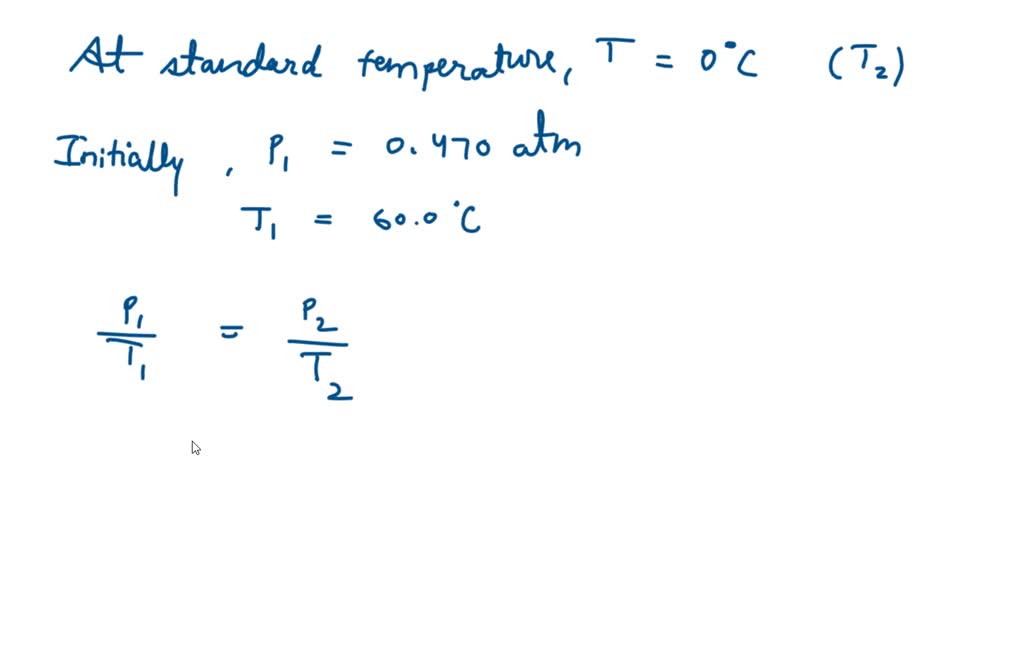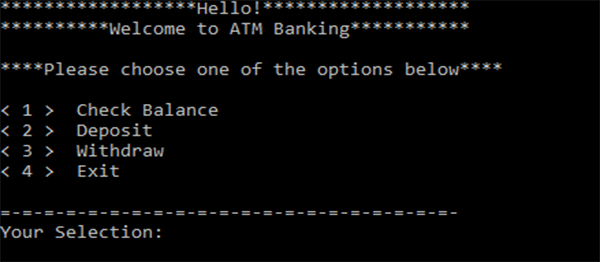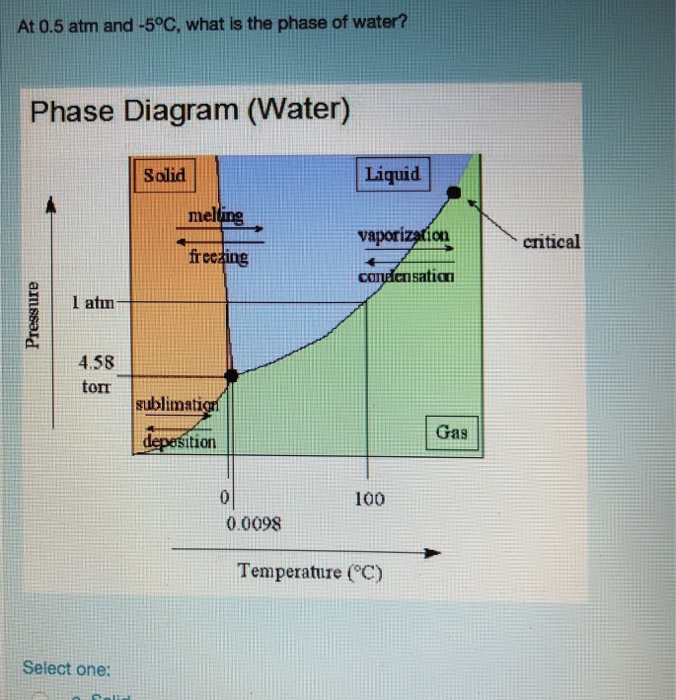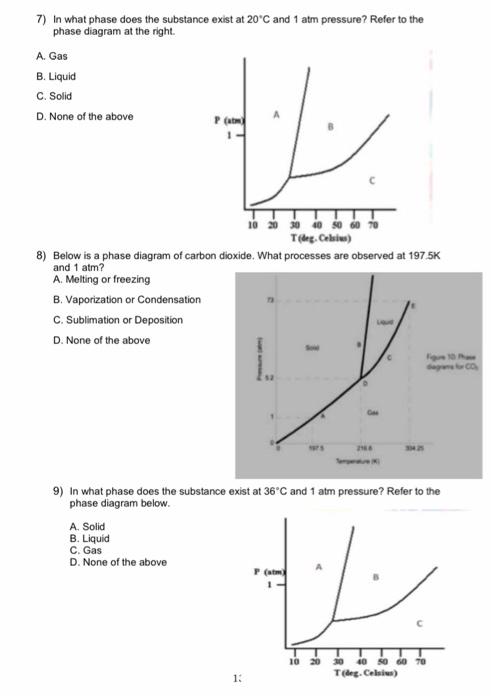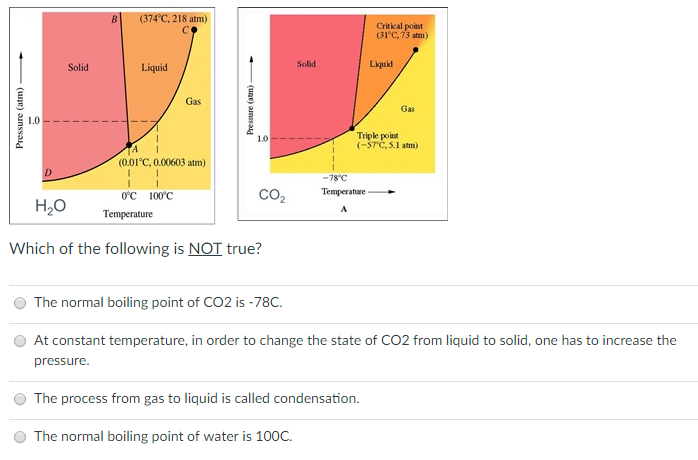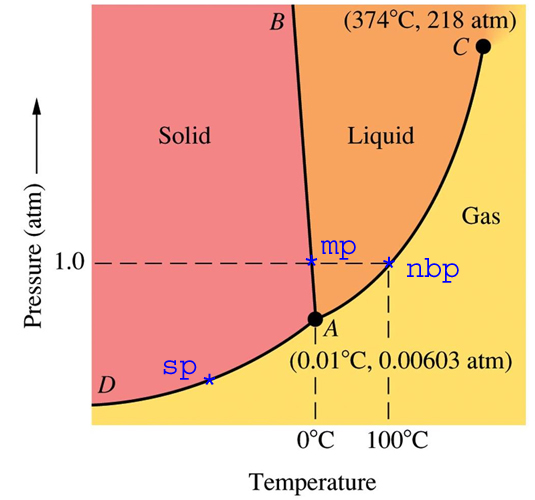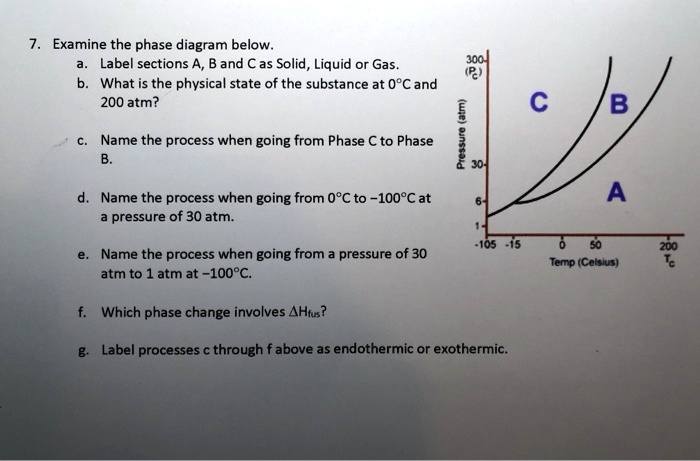
SOLVED: Examine the phase diagram below Label sections A, B and C as Solid, Liquid or Gas. 300 What is the physical state of the substance at 0"C and 200 atm? Name

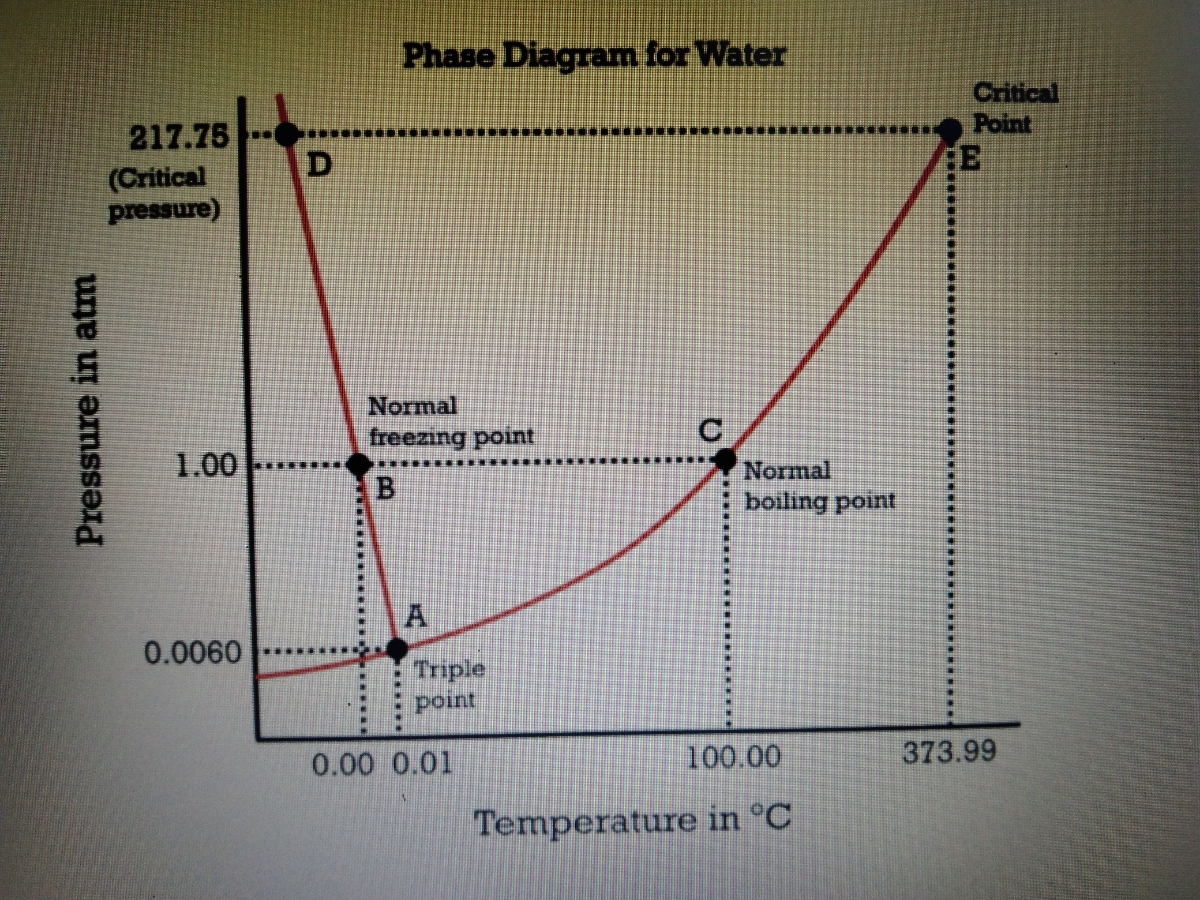
Which phase/s of water exist uder 0.35 atm and 71 degC? a. solid b. liquid c. gas d. supercritical fluid | Homework.Study.com

Gas Pressure Unit Conversions - torr to atm, psi to atm, atm to mm Hg, kpa to mm Hg, psi to torr - YouTube

SOLVED:Helium gas in a 2.00-L cylinder is under 1.12 atm pressure. At 36.5°C, that same gas sample has a pressure of 2.56 atm. What was the initial temperature of the gas in

In what phase is CO2 at 4 atm and -10 degrees Celsius? Is it a liquid, gas, or solid? | Homework.Study.com
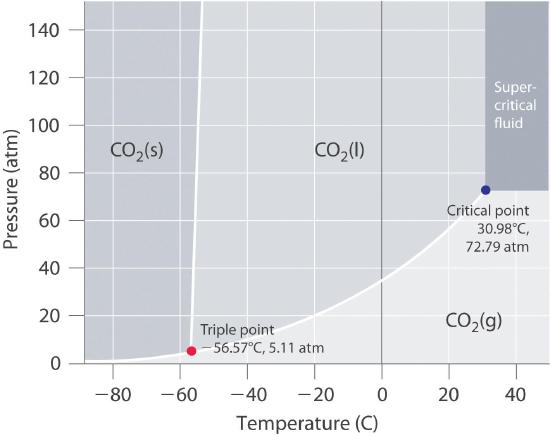
Using the phase diagram for CO_2, what phase is carbon dioxide in at -20 C and 1 atm pressure? | Socratic
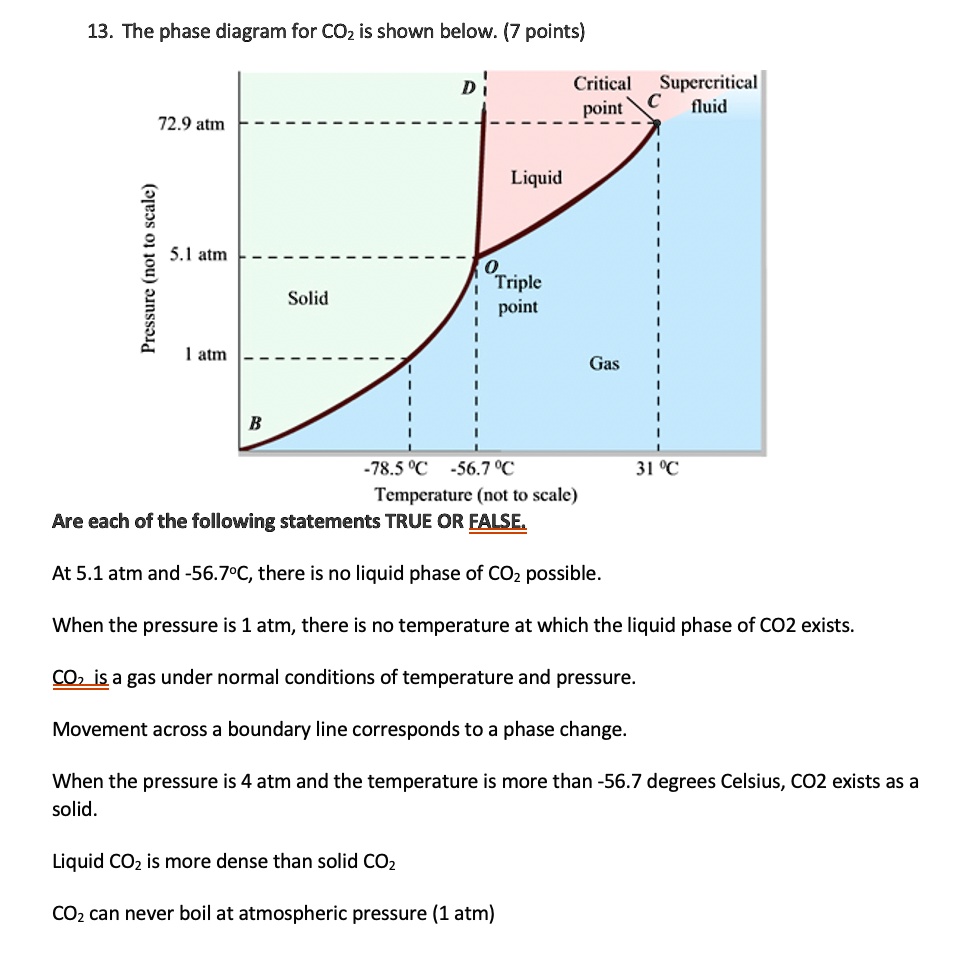
SOLVED: 13 The phase diagram for COz is shown below: points) Critical point Supereritical fluid 72.9 atm Liquid 1 1 5.1 atm L aln Triple point Solid Gas 78.5 %C 56.7 "€

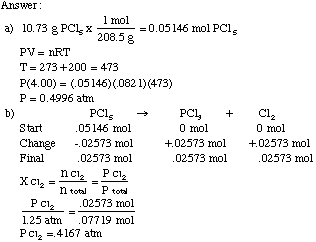
SOLVED: Part A The pressure inside hydrogen-filled container was 2.10 atm at 21 C. What would the pressure be if the container was heated to 95 "C ? Express your answer with