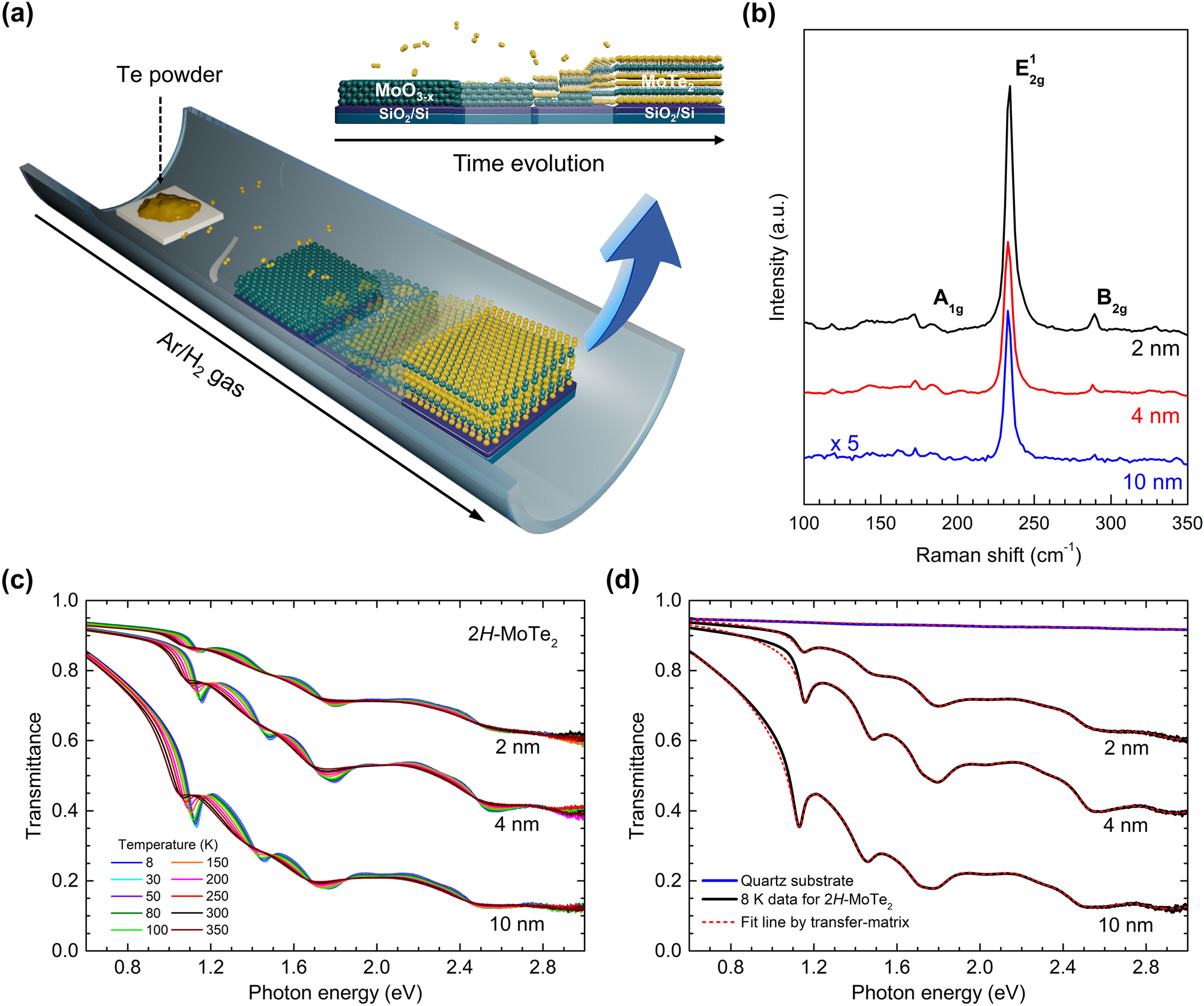
Highly Efficient Synthesis of Carbon-Based Molybdenum Phosphide Nanoparticles for Electrocatalytic Hydrogen Evolution | Nanoscale Research Letters | Full Text
Molybdenum Disulphide Nanoparticles Synthesis Using a Low Temperature Hydrothermal Method and Characterization

Generalized molybdenum oxide surface chemical state XPS determination via informed amorphous sample model - ScienceDirect

In depth study of molybdenum silicon compound formation at buried interfaces: Journal of Applied Physics: Vol 120, No 11
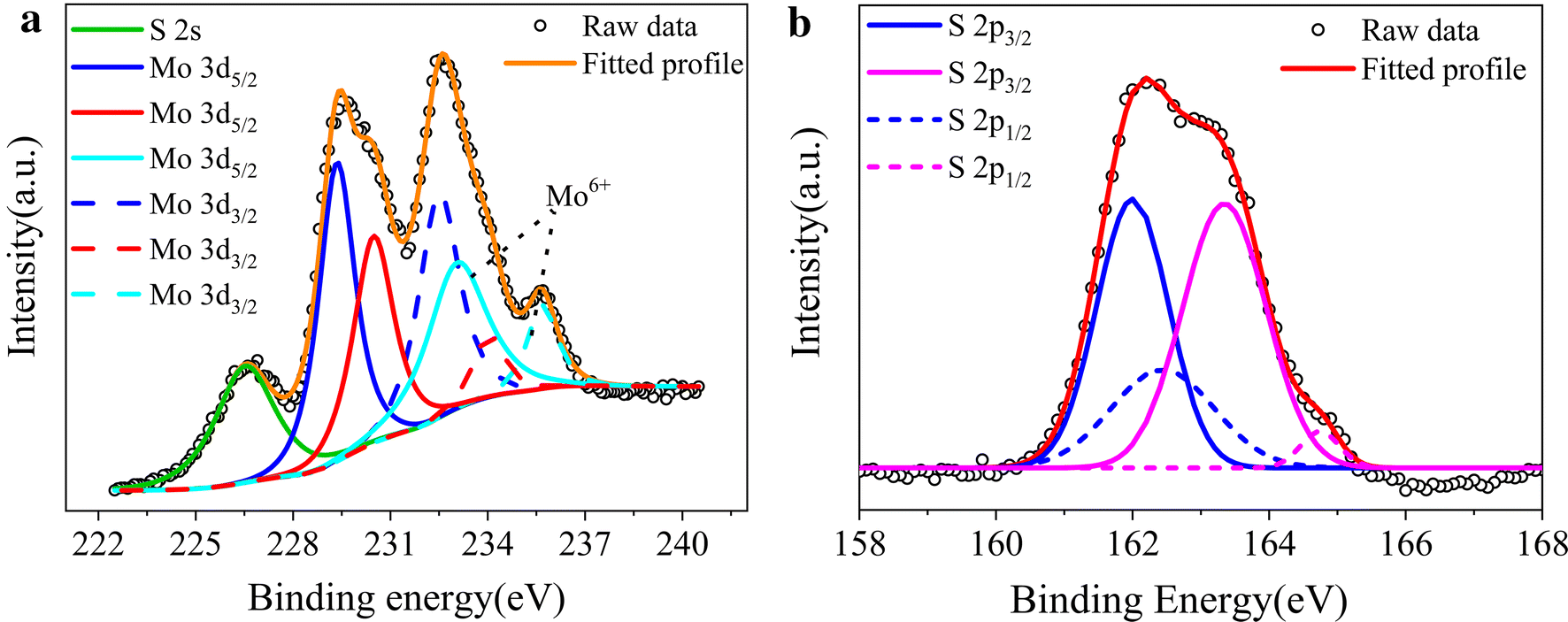
Electrodeposition of amorphous molybdenum sulfide thin film for electrochemical hydrogen evolution reaction | BMC Chemistry | Full Text
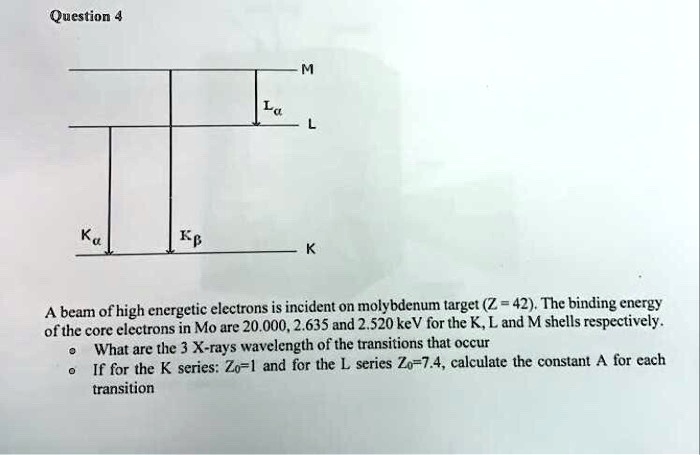
SOLVED: Question 4 Ku A beam of high energetic electrons is incident on molybdenum target (Z =.42) The binding energy of the core clectrons in Mo are 20.000,2.635 and 2.520 keV for
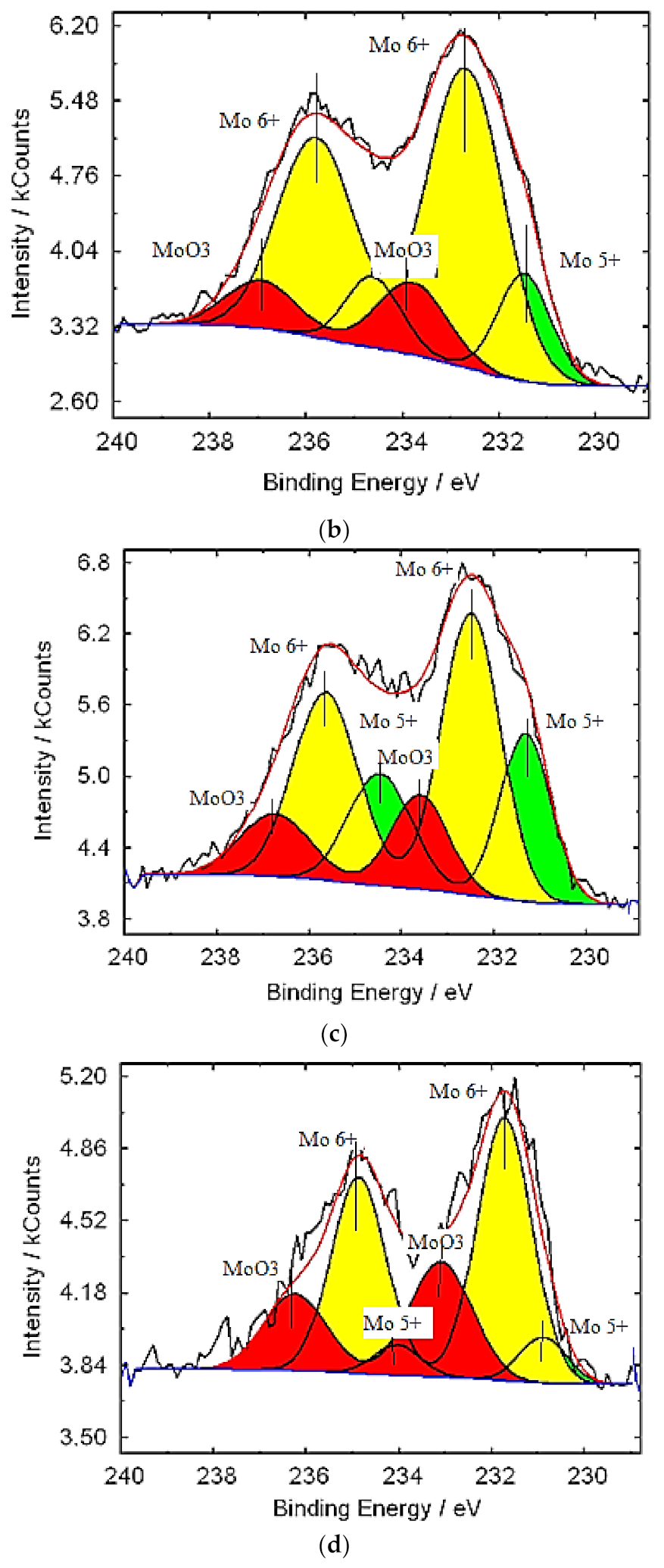





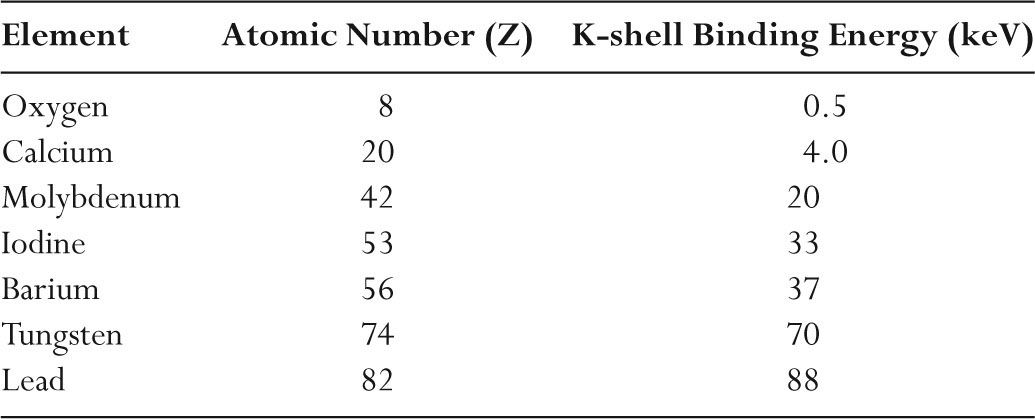



![PDF] Oxidation of molybdenum by low-energy oxygen-ion bombardment | Semantic Scholar PDF] Oxidation of molybdenum by low-energy oxygen-ion bombardment | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/8399cdd43669cf9a6e553b0f32df54feed64b428/3-Figure4-1.png)