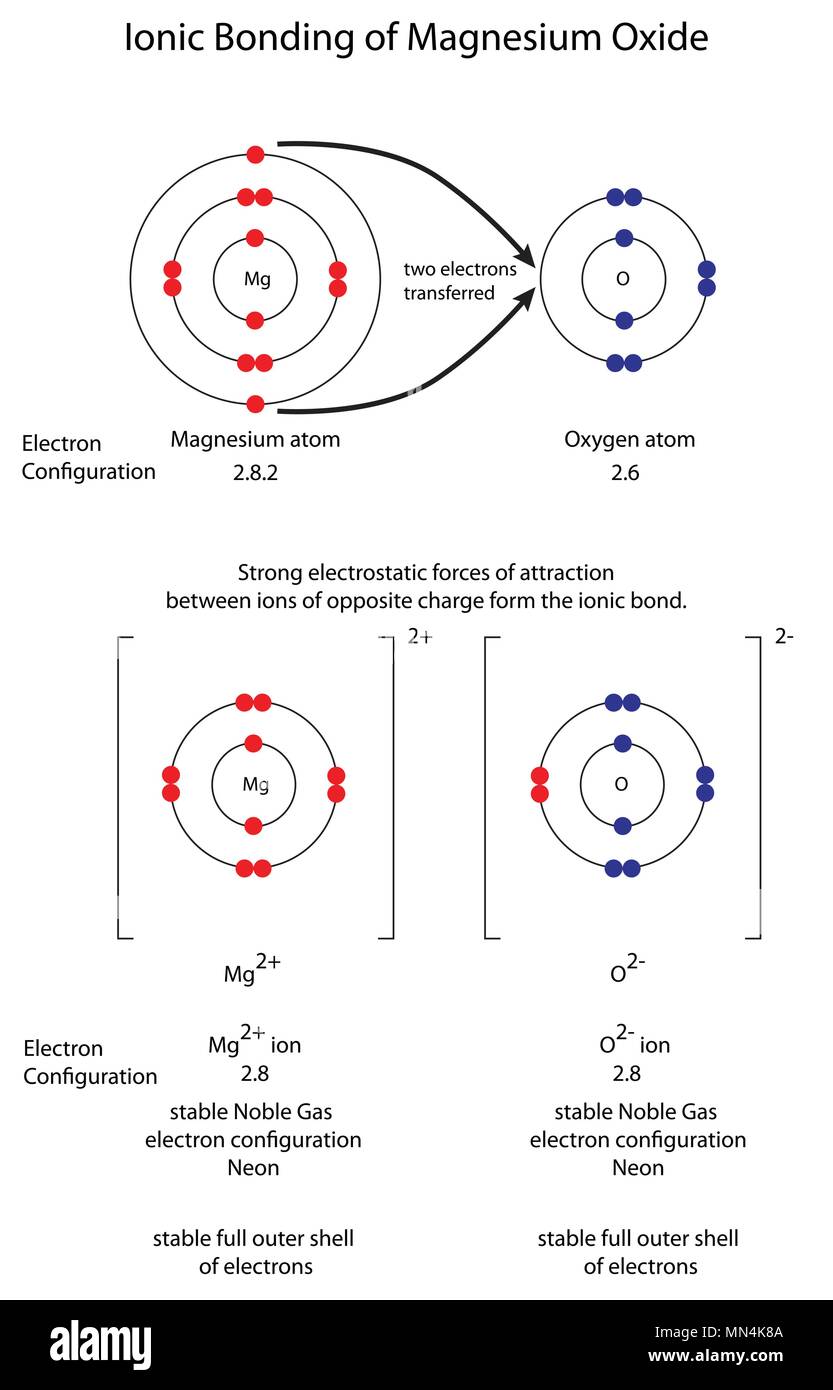
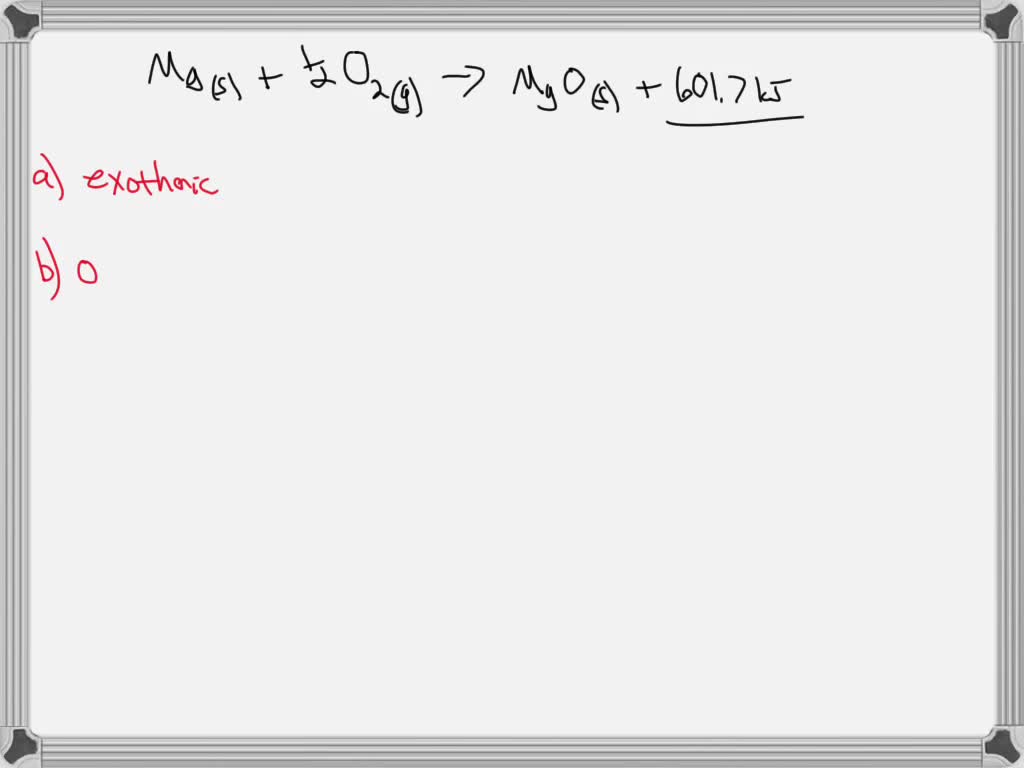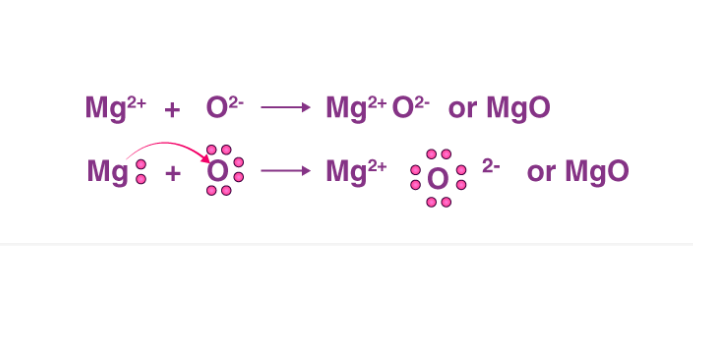Show the formation of MgO by the transfer of electrons in the two elements - CBSE Class 10 Science - Learn CBSE Forum
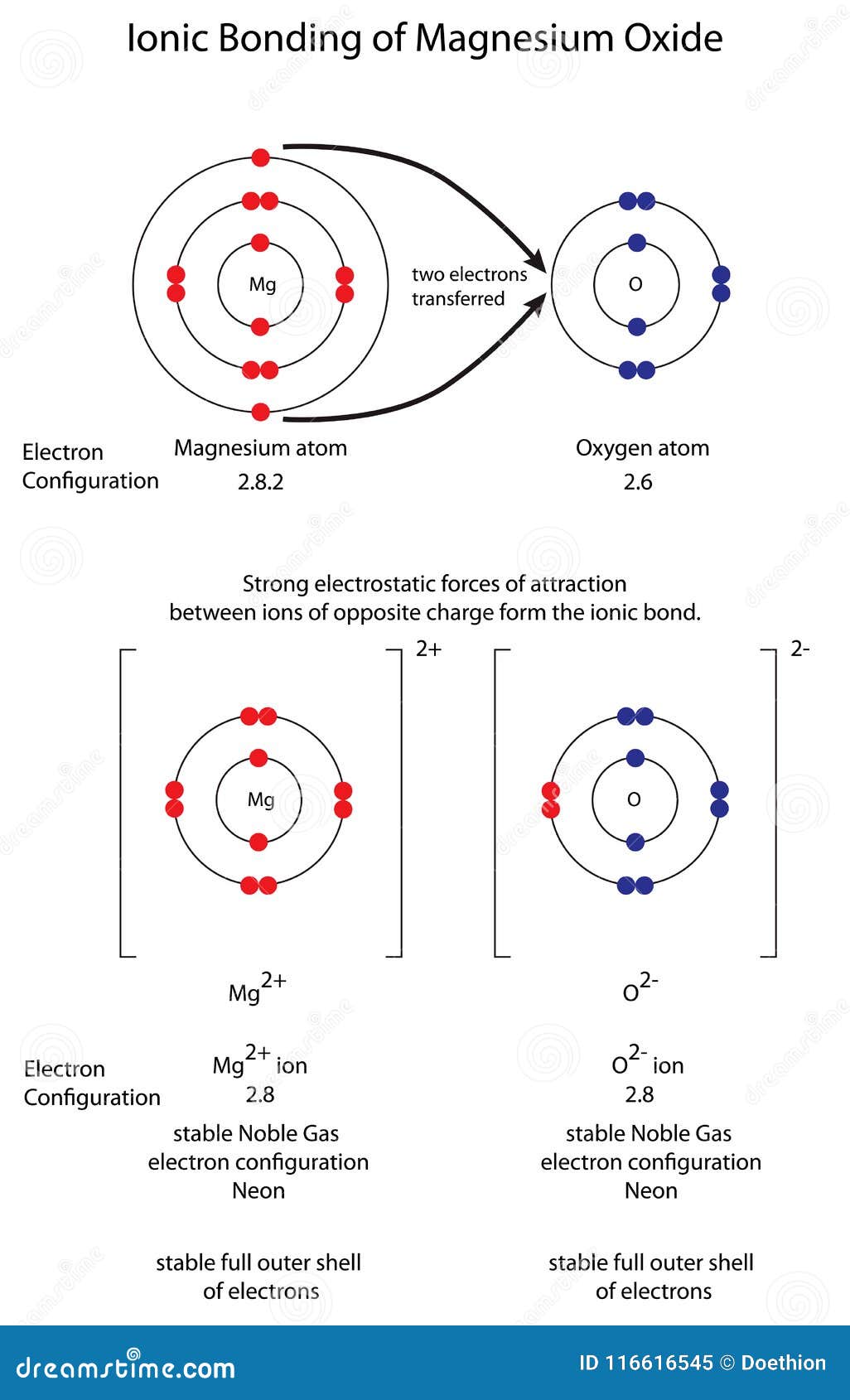
Diagram To Show Ionic Bonding in Magnesium Oxide MgO Stock Illustration - Illustration of compound, formation: 116616545
Experiment 7: Enthalpy of Formation of Magnesium Oxide Purpose: In this experiment, Hess' law will be used to determine the ent

SOLVED:The enthalpy of formation of magnesium oxide, MgO(s), is -601827 kJ / kmol at 25^∘ C. The melting point of magnesium oxide is approximately 3000 K, and the increase in enthalpy between

Enthalpy of Formation of Magnesium Oxide - Experiment 9: Enthalpy of Formation of Magnesium Oxide - Studocu