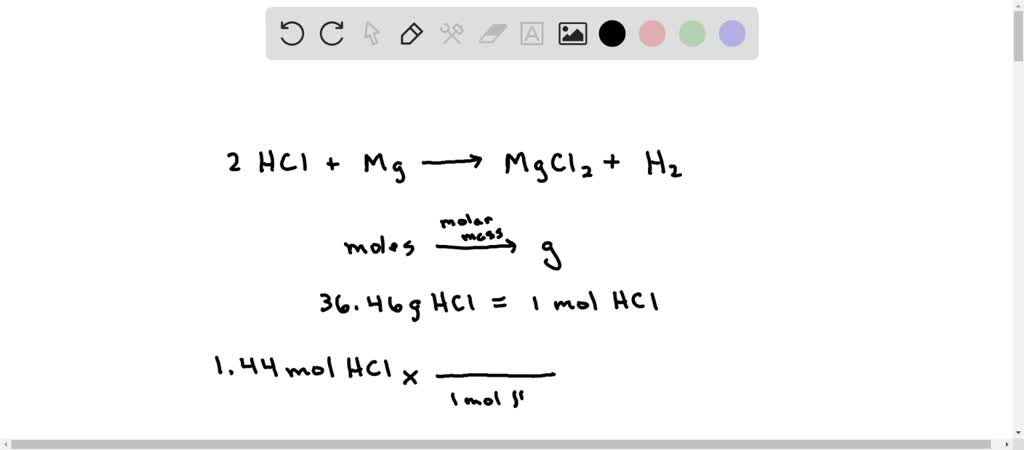
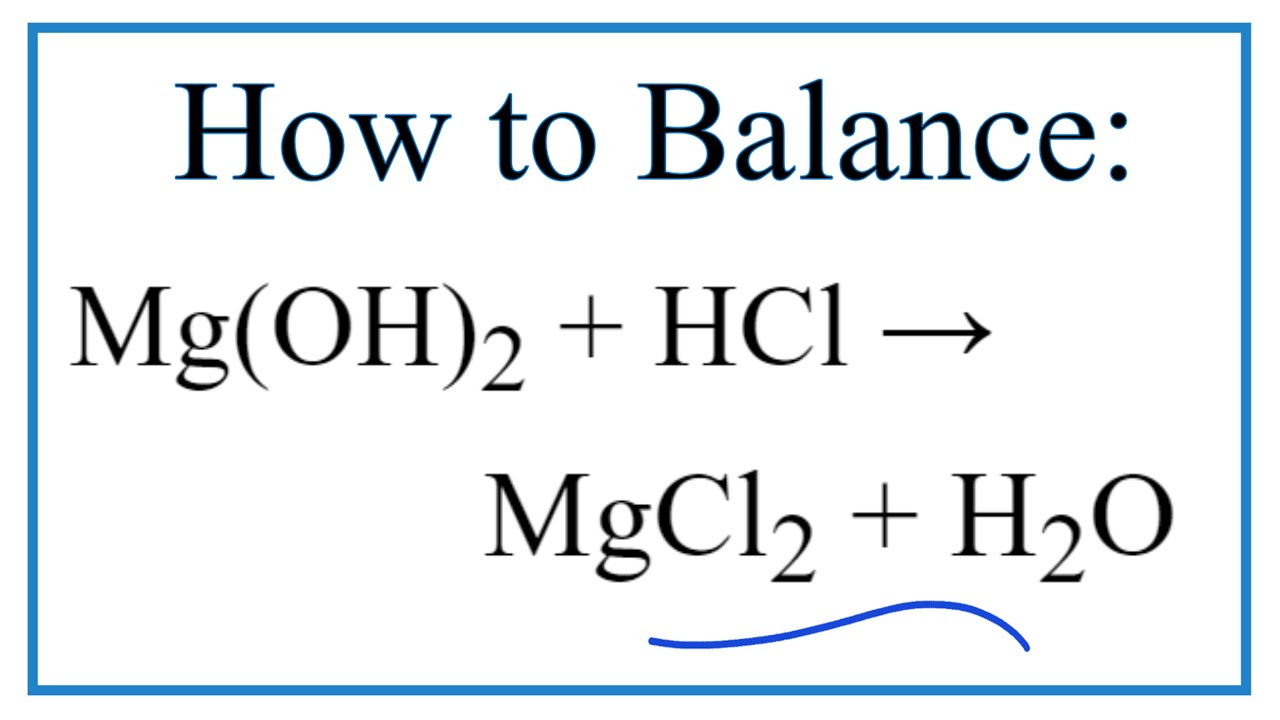
SOLVED: When hydrochloric acid reacts with magnesium hydroxide, the products that form are magnesium chloride and water. What is the coefficient for water when this equation is properly balanced?

Question Video: Recognizing Why Warm Hydrochloric Acid Has a Faster Rate of Reaction with Magnesium than Cold Hydrochloric Acid | Nagwa

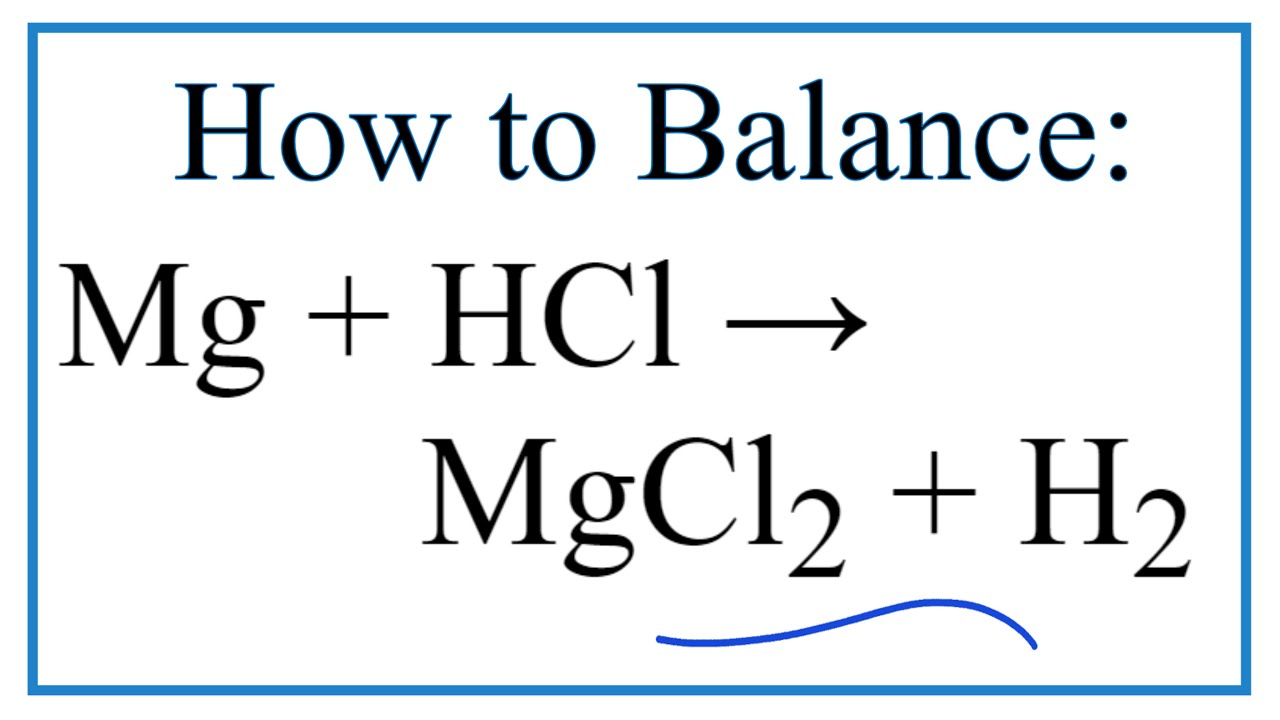
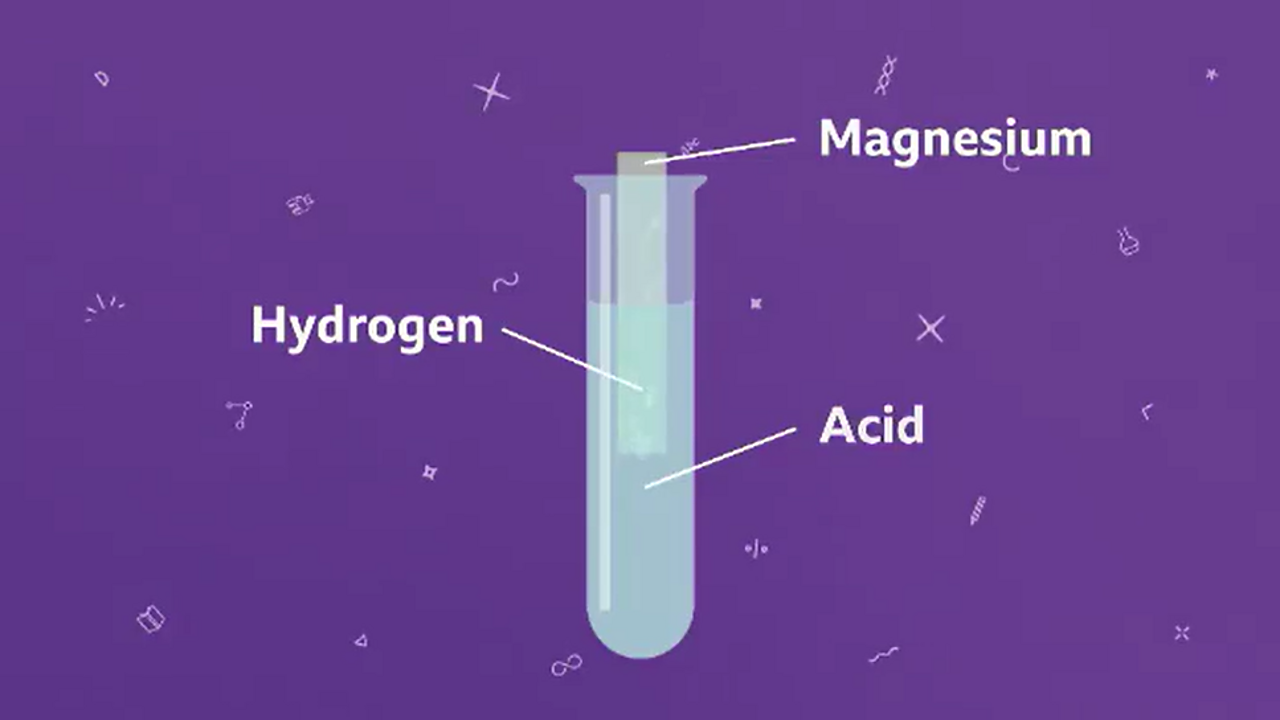
How to Balance Mg + HCl → MgCl2 + H2 (Magnesium + Hydrochloric Acid) | How to Balance Mg + HCl → MgCl2 + H2 (Magnesium + Hydrochloric Acid) One of the

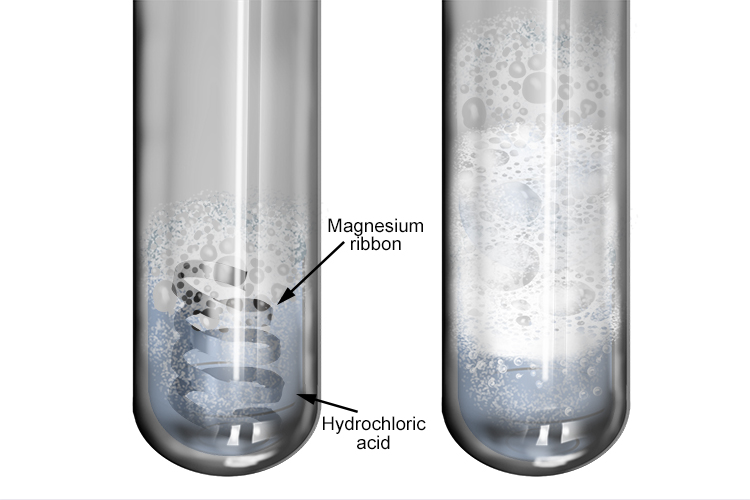
What happens magnesium react with hydrochloric acid? - Find 3 Answers & Solutions | LearnPick Resources