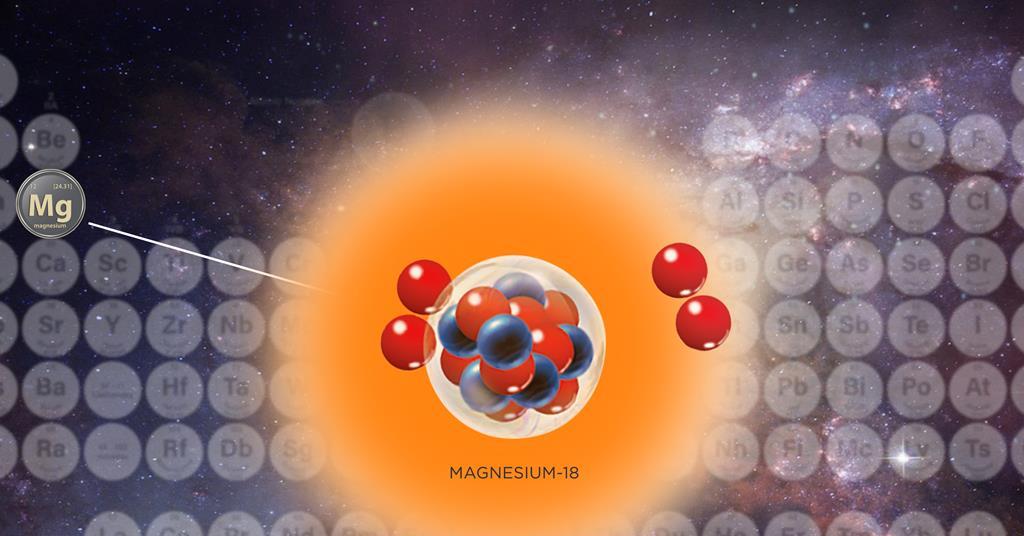
Shortest-lived and lightest magnesium isotope ever too unstable to even attract electrons | Research | Chemistry World
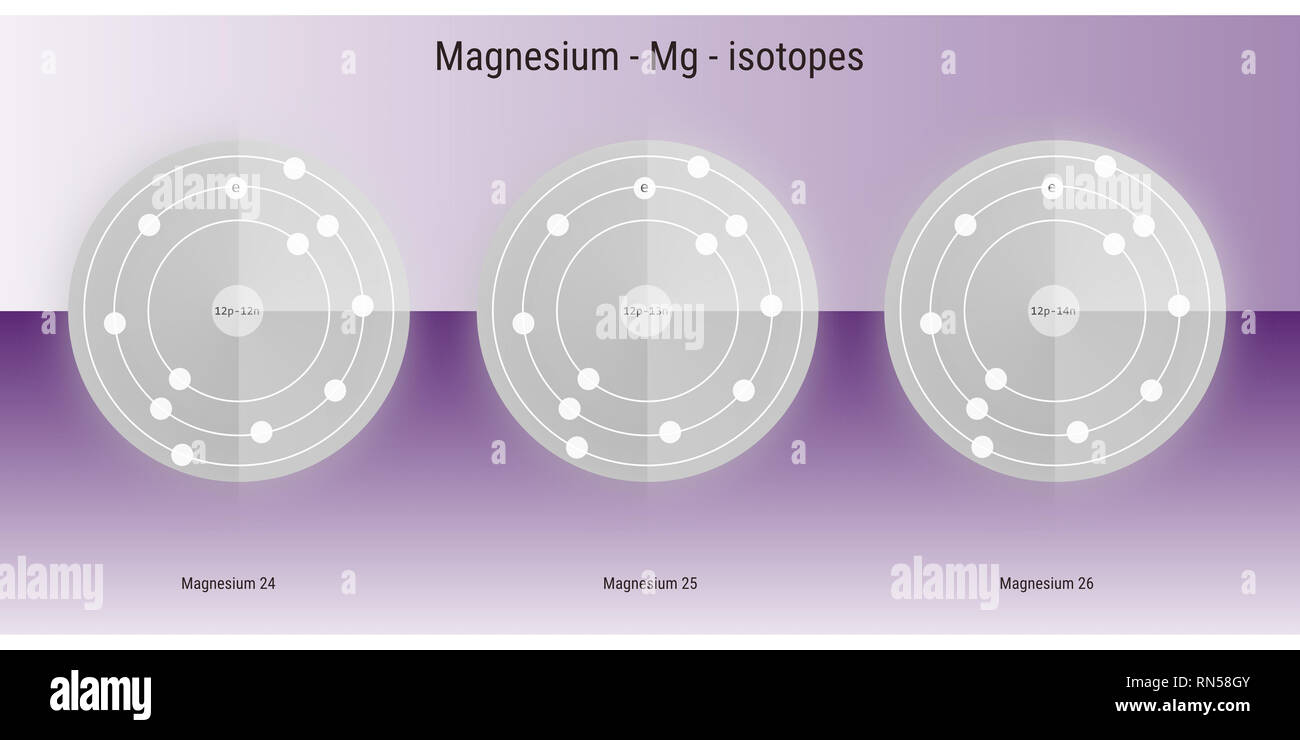
magnesium isotopes atomic structure backdrop - physics theory illustration schematic Stock Photo - Alamy

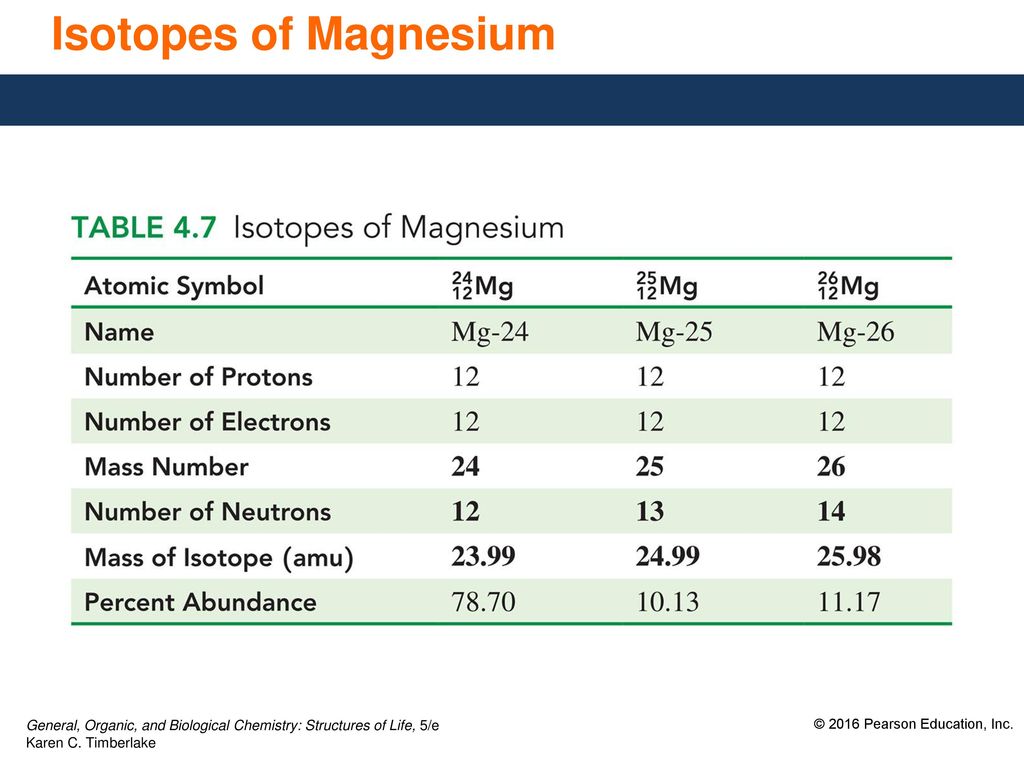
Magnesium has two isotopes ^2412Mg and ^2612Mg .Both the isotops have same electronic configurations.
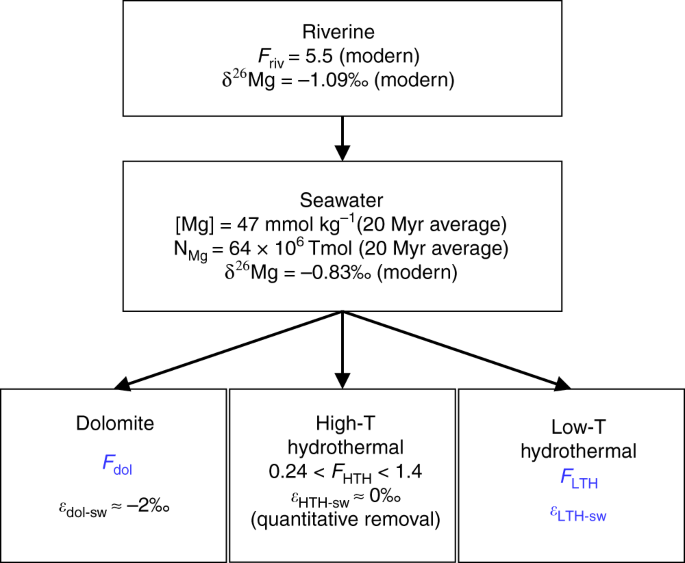
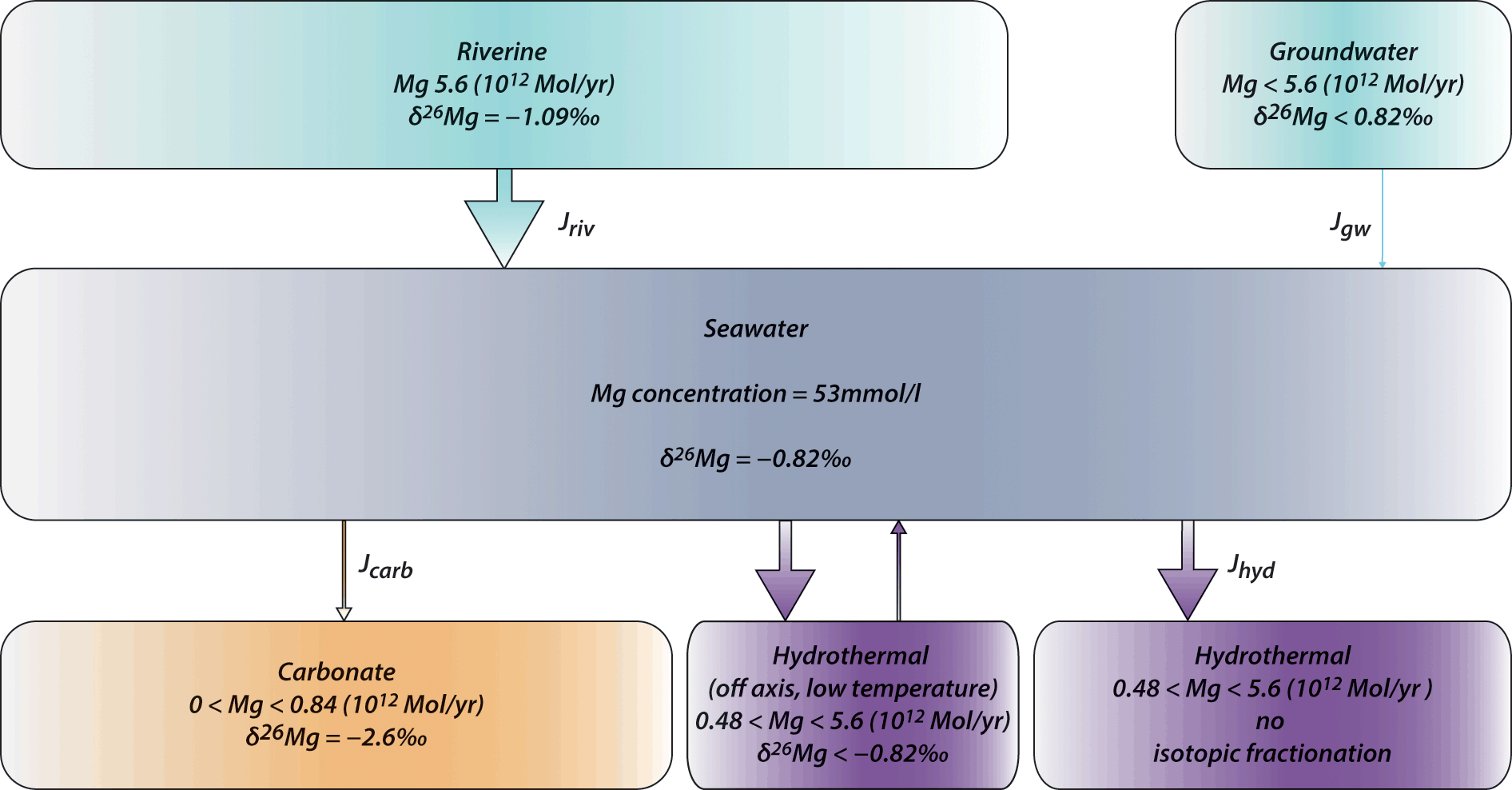
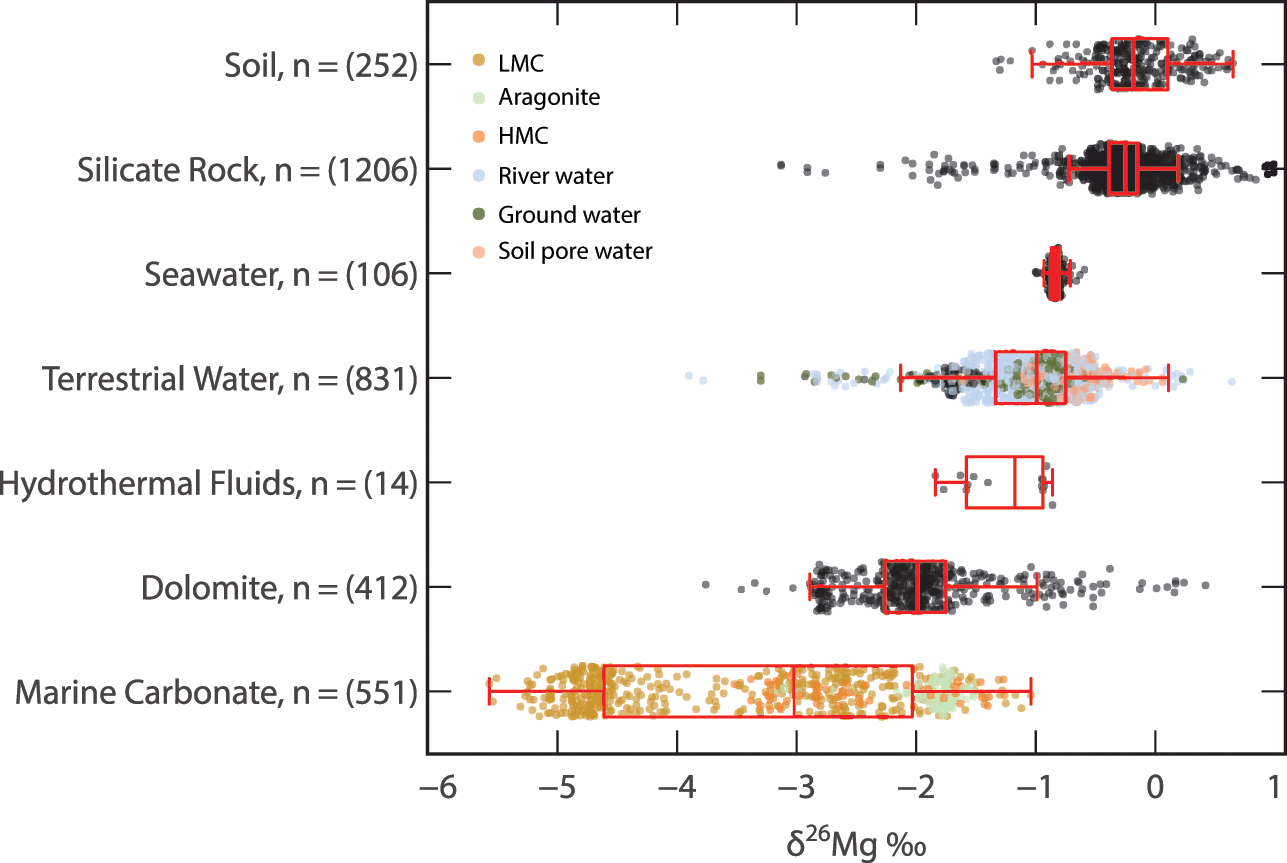
New isotope constraints on the Mg oceanic budget point to cryptic modern dolomite formation | Nature Communications
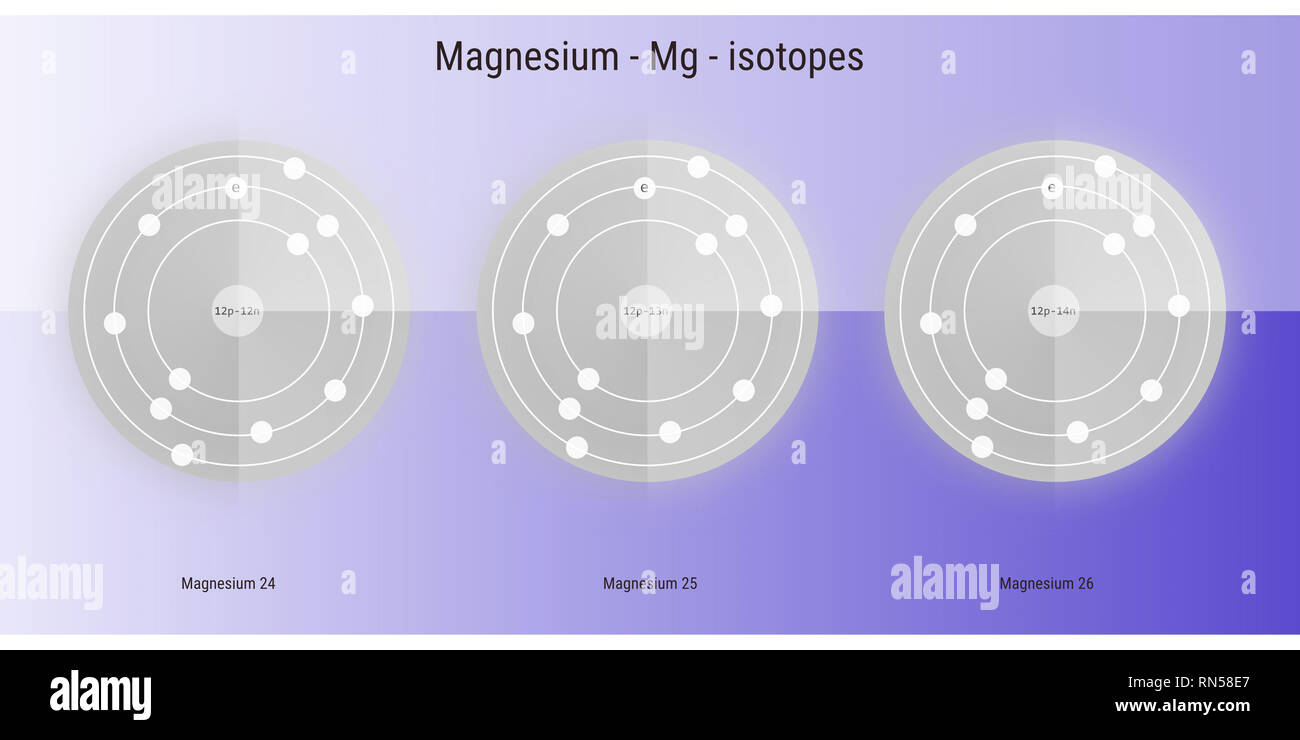
magnesium isotopes atomic structure backdrop - physics theory illustration schematic Stock Photo - Alamy

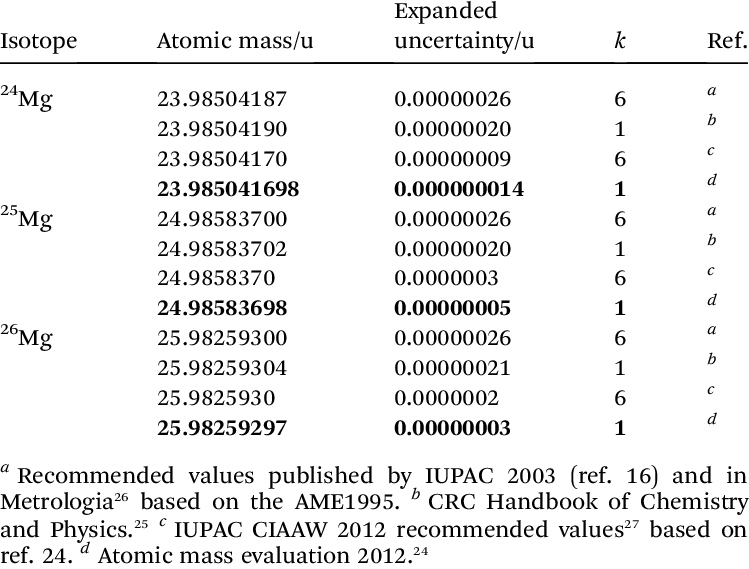
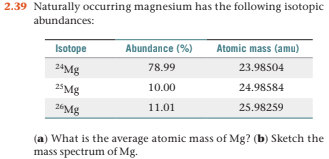
Magnesium has three naturally occurring isotopes with the following masses and natural abundances: \begin{array}{|c|c|c|} \hline \text{Isotope} & \text{Mass (amu)} & \text{Abundance (%)} \\ \hline \text{Mg-24} & \text{23.9850} & \text{78.99} \\ \hline \t

magnesium isotopes atomic structure backdrop - physics theory illustration schematic Stock Photo - Alamy

Magnesium has two isotopes ^2412Mg and ^2612Mg . According to which isotopes of magnesium have different mass numbers.

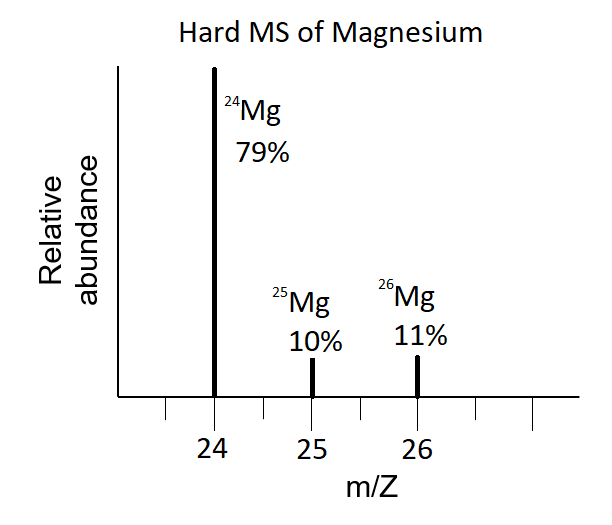
SOLVED: Magnesium has three naturally occurring isotopes with masses of 24, 25,26 a.m.u and natural abundances of 79%, 10% and 11%. Calculate the atomic mass of magnesium in U or Da; Sketch

Magnesium isotopes tracer global biogeochemical cycle magnesium past and present or archive alteration | Geochemistry and environmental chemistry | Cambridge University Press