What is an oxide? An oxide is a Binary compound of oxygen and another element. M & O Oxides can be classified in two ways – Nature of Oxides Amount of. -

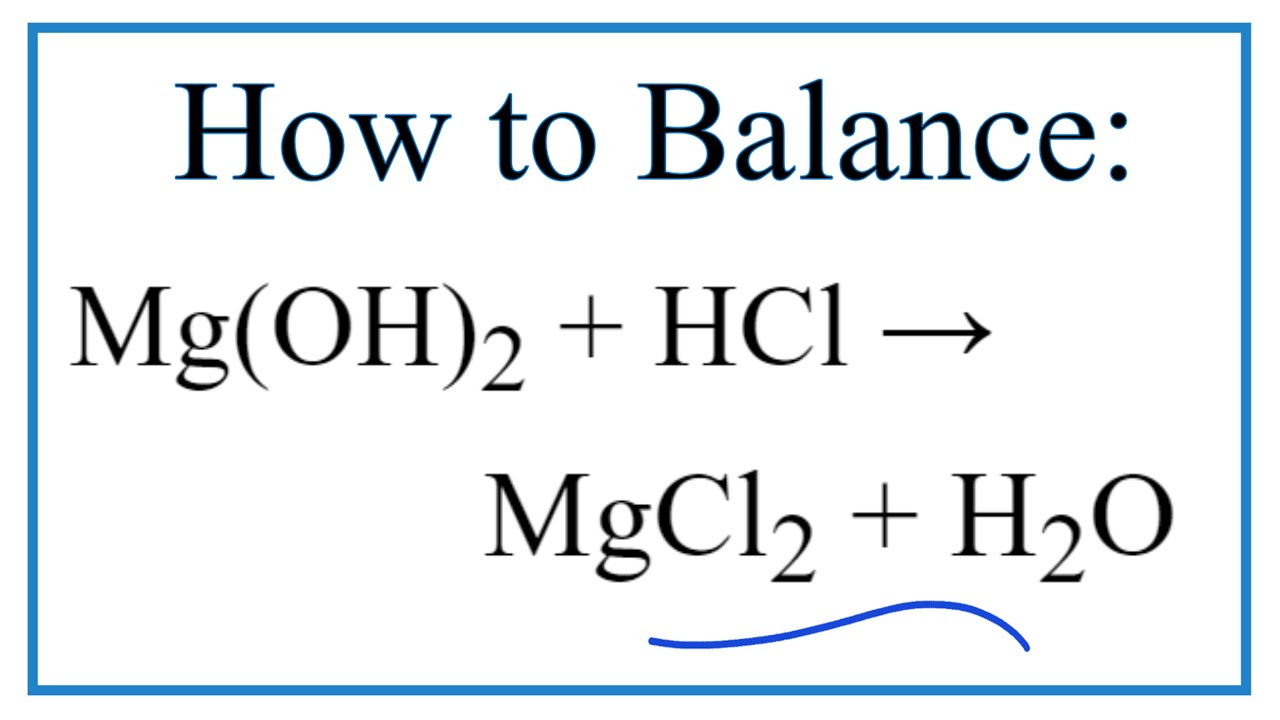
SOLVED:Magnesium carbonate, magnesium oxide, and magnesium hydroxide are all white solids that react with acidic solutions. (a) Write a balanced molecular equation and a net ionic equation for the reaction that occurs

Write the balanced chemical equations for the following reaction:Calcium hydroxide + Carbon dioxide → Calcium Carbonate + Water.

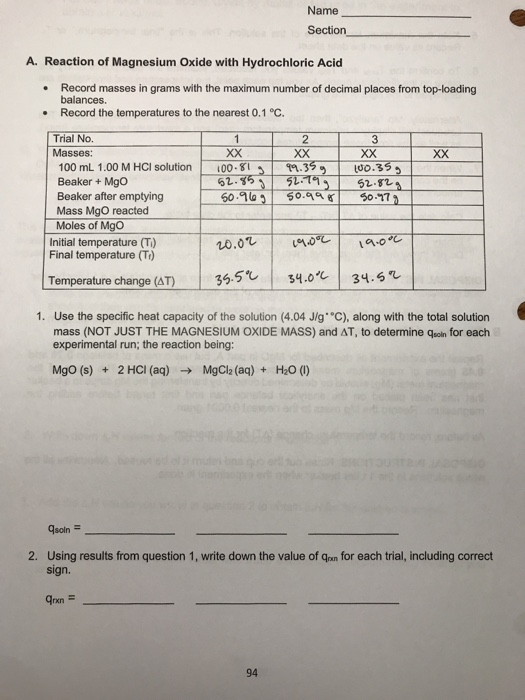
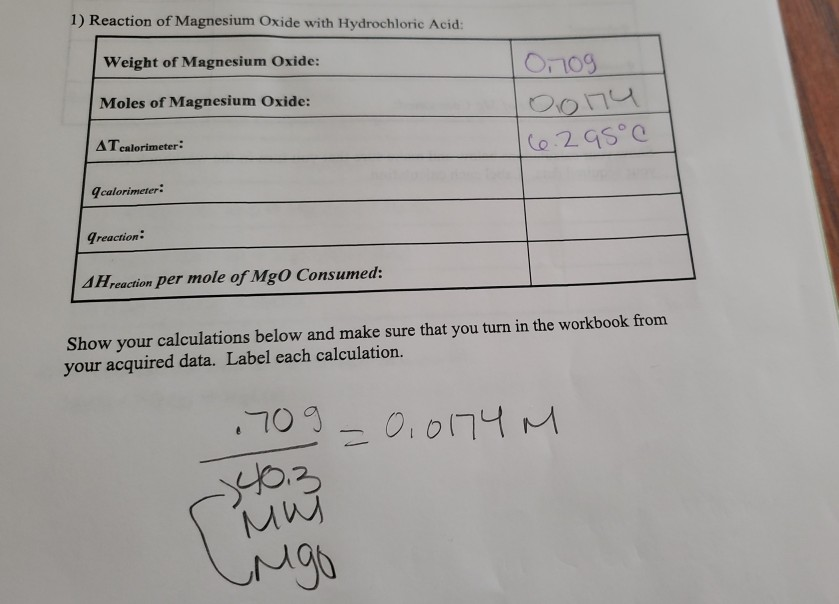
SOLVED: Reaction of Magnesium Oxide with Hydrochloric Acid Trial No Masses: XX XX 100 mL 00 M HCI solution Jno Ghs 91. 5S4 1o.31 Beaker Mgo 52 629 52 ,219 S2,6L) Beaker