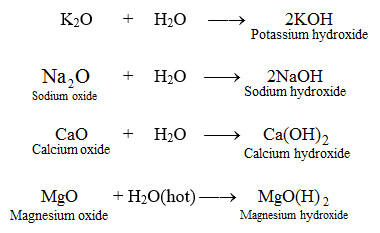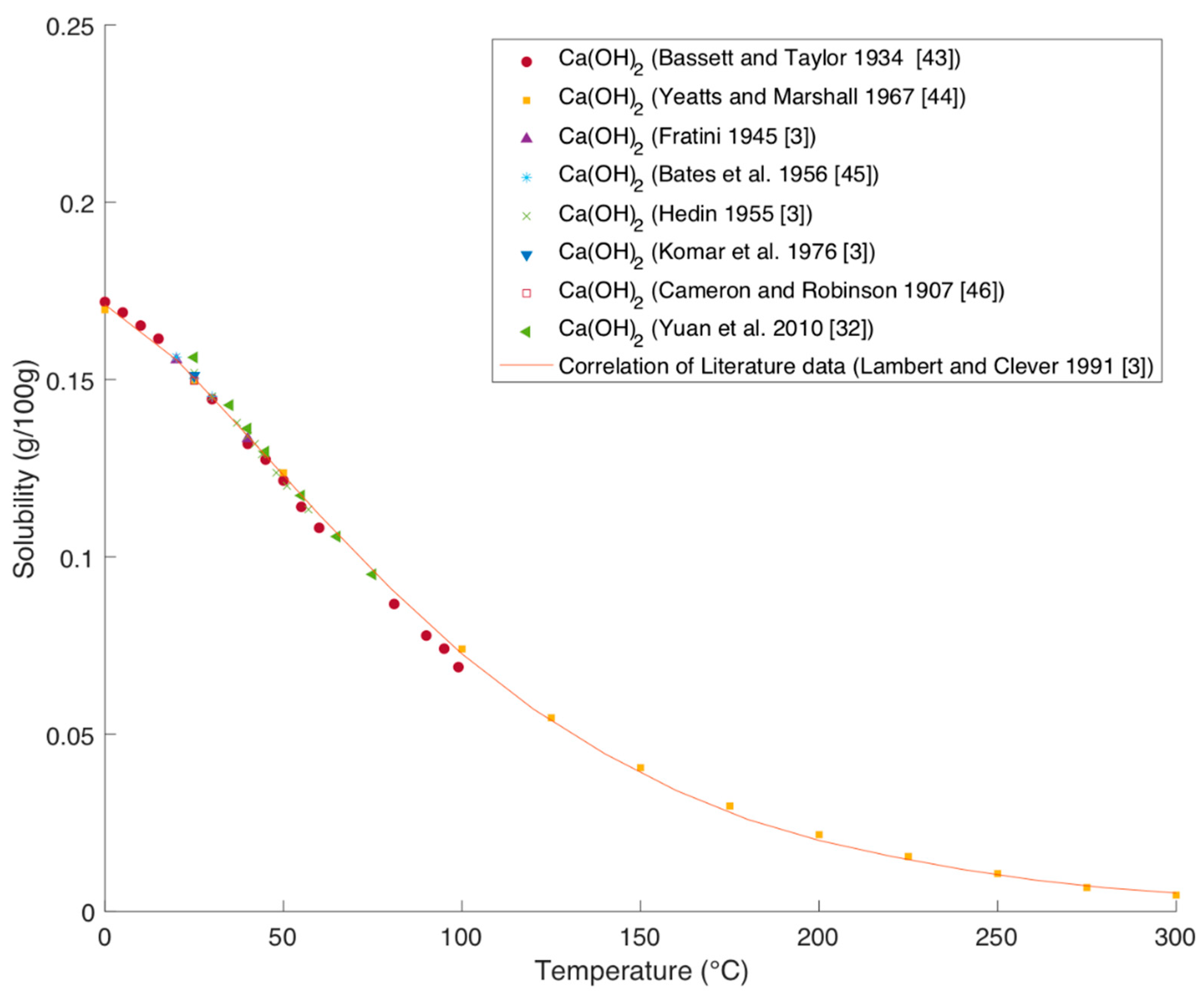
Processes | Free Full-Text | Solubility Data of Potential Salts in the MgO-CaO-SO2-H2O-O2 System for Process Modeling
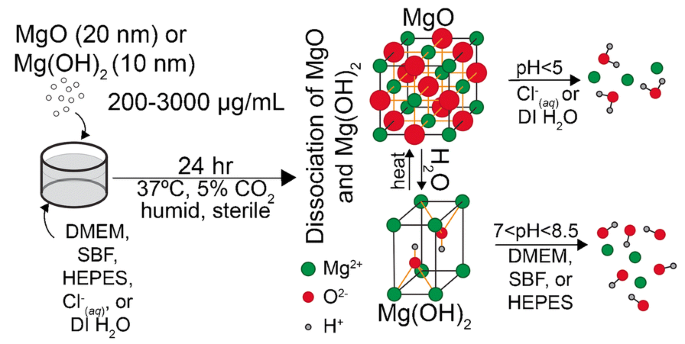
Dissociation of magnesium oxide and magnesium hydroxide nanoparticles in physiologically relevant fluids | SpringerLink

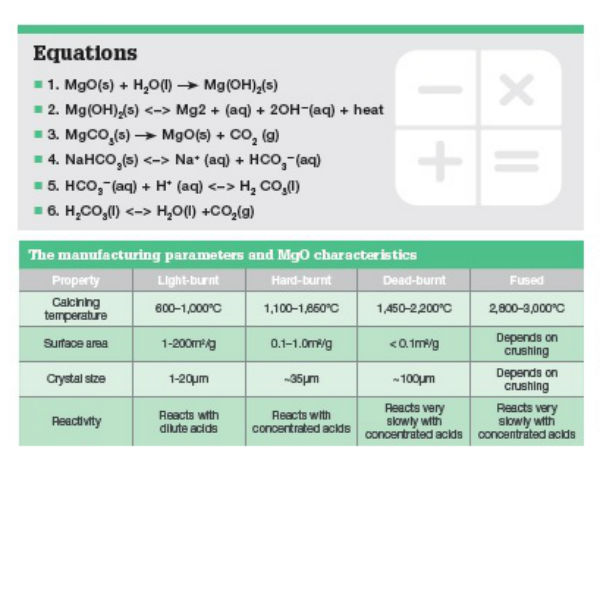
THE HYDRATION OF MAGNESIUM OXIDE WITH DIFFERENT REACTIVITIES BY WATER AND MAGNESIUM ACETATE | Semantic Scholar

The solubility of magnesium sulphite hydrates calculated by Aspen Plus®... | Download Scientific Diagram

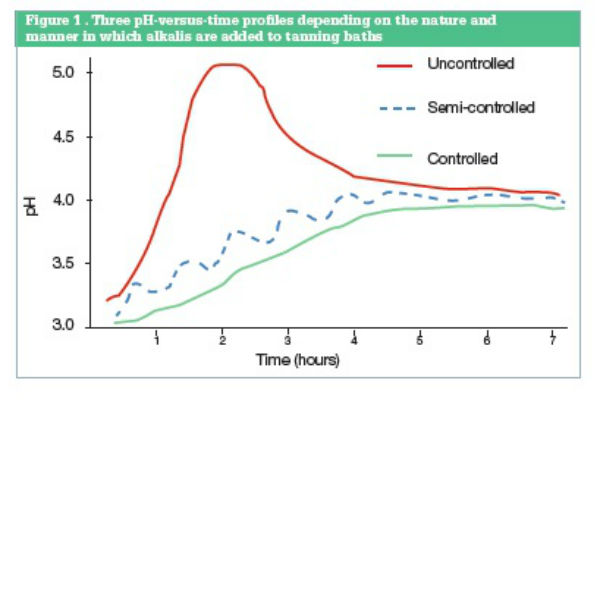
Prediction of adequate pH and Mg2+ dosage using an empirical MgO solubility model for struvite crystallization - ScienceDirect

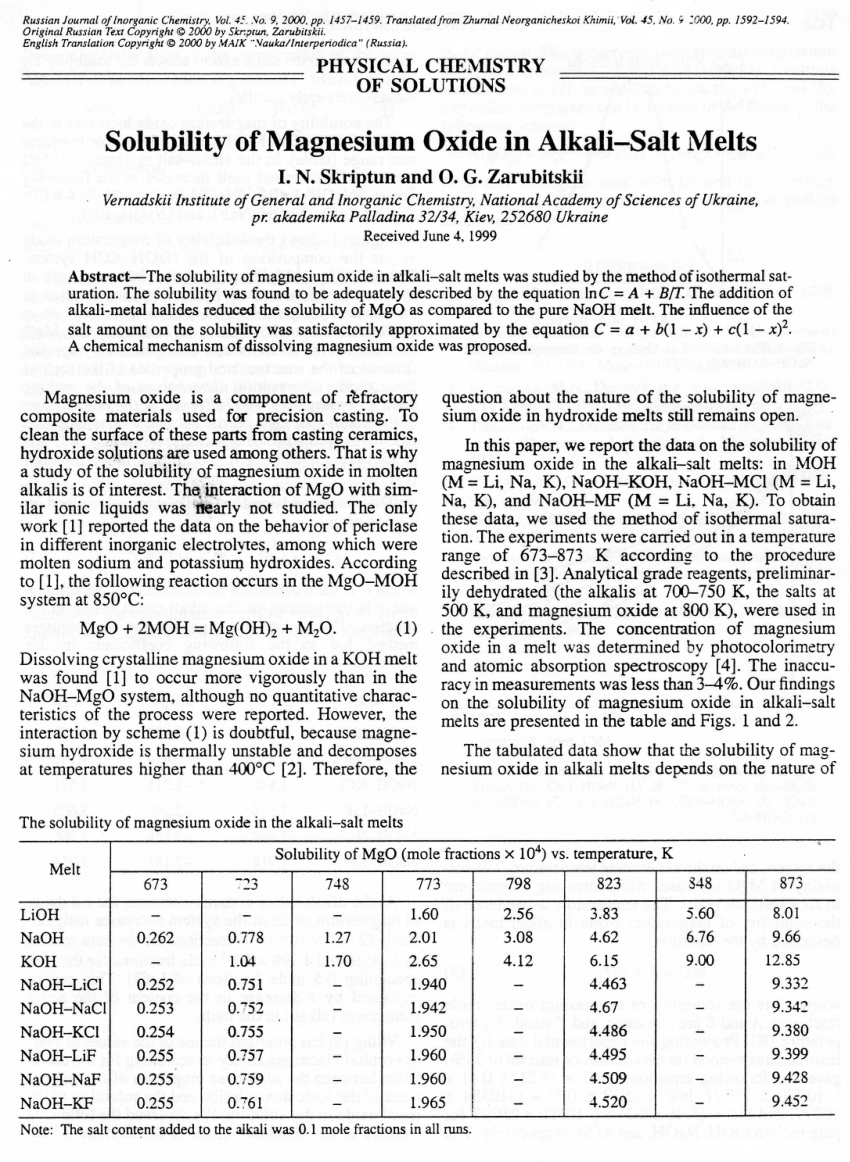
On some regularities of magnesium oxide solubility in melts with different content of alkaline earth metal chlorides - ScienceDirect

Dissociation of magnesium oxide and magnesium hydroxide nanoparticles in physiologically relevant fluids | SpringerLink