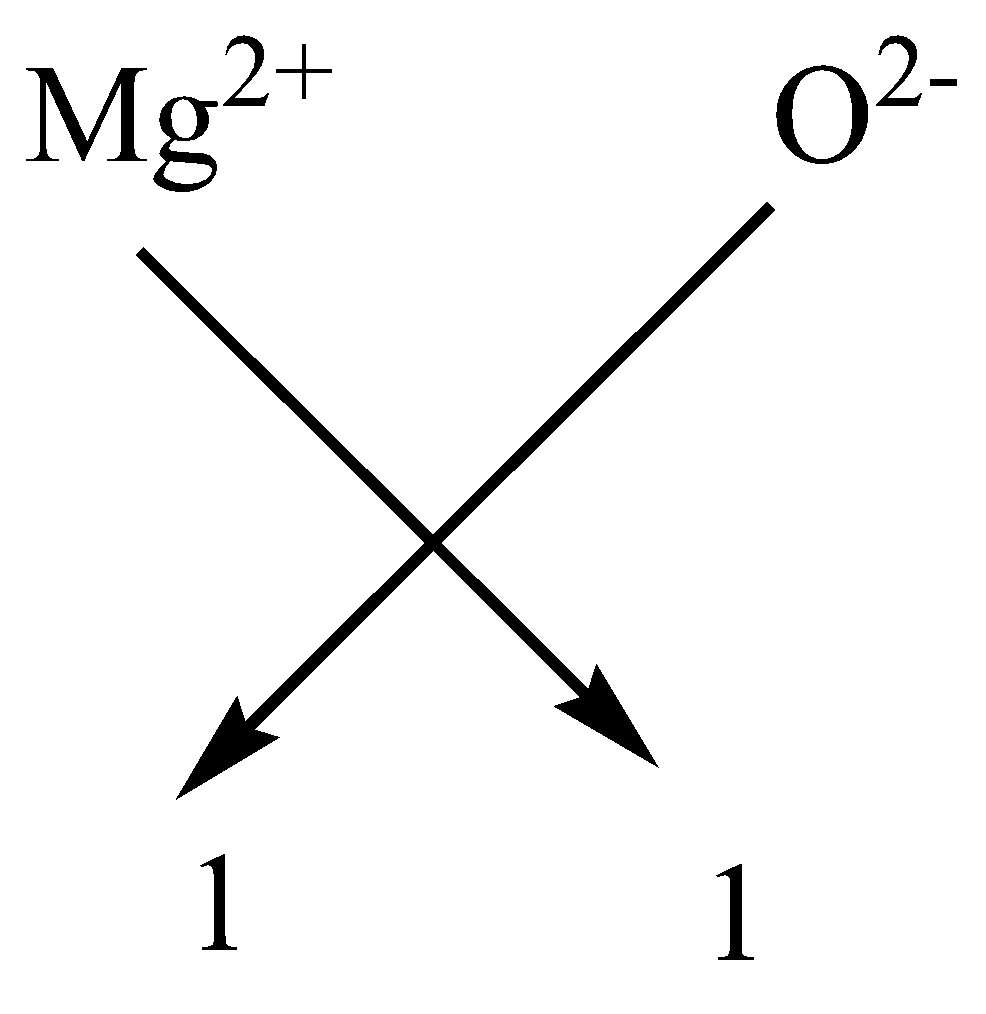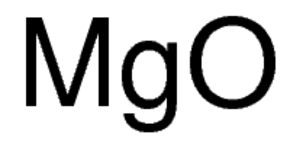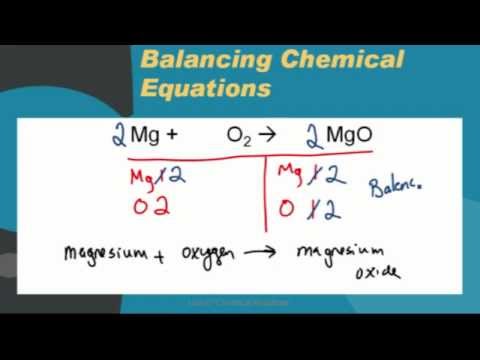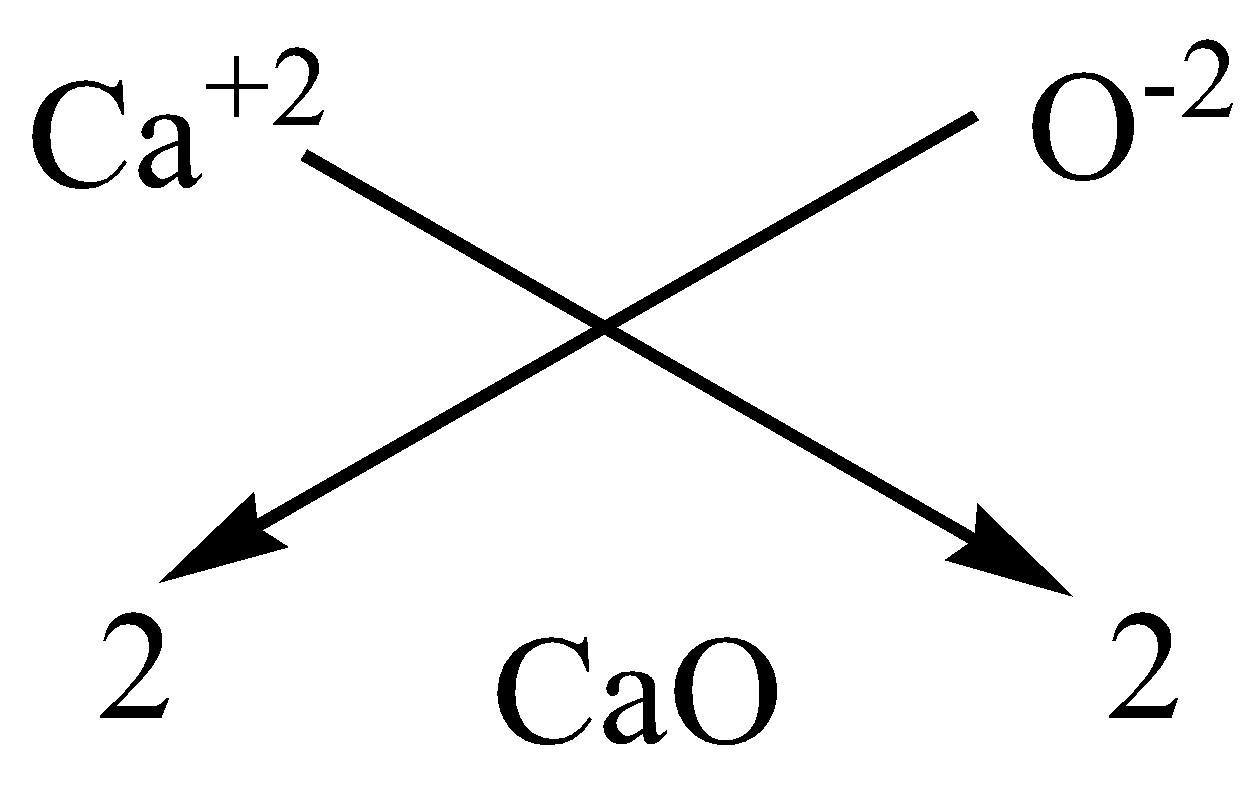
Write the chemical formulae of the following by criss-cross method:A. Magnesium chlorideB.Calcium oxideC.Copper nitrateD.Aluminium chlorideE.Potassium nitrate
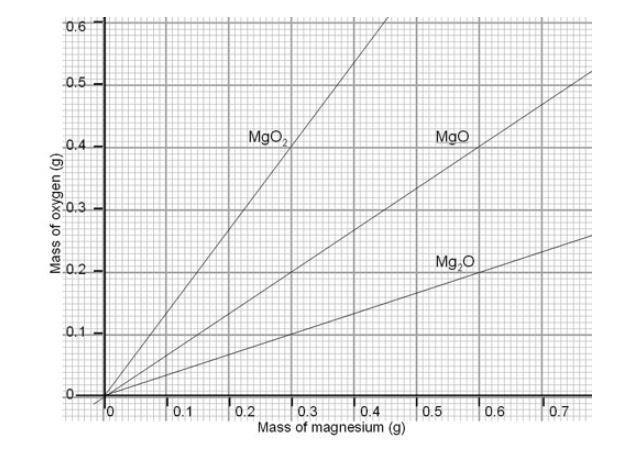
A 3.250g sample of magnesium is burned in a container of 12.500g oxygen. What mass of oxygen gas remains unreacted after the magnesium has been completely consumed to form magnesium oxide as
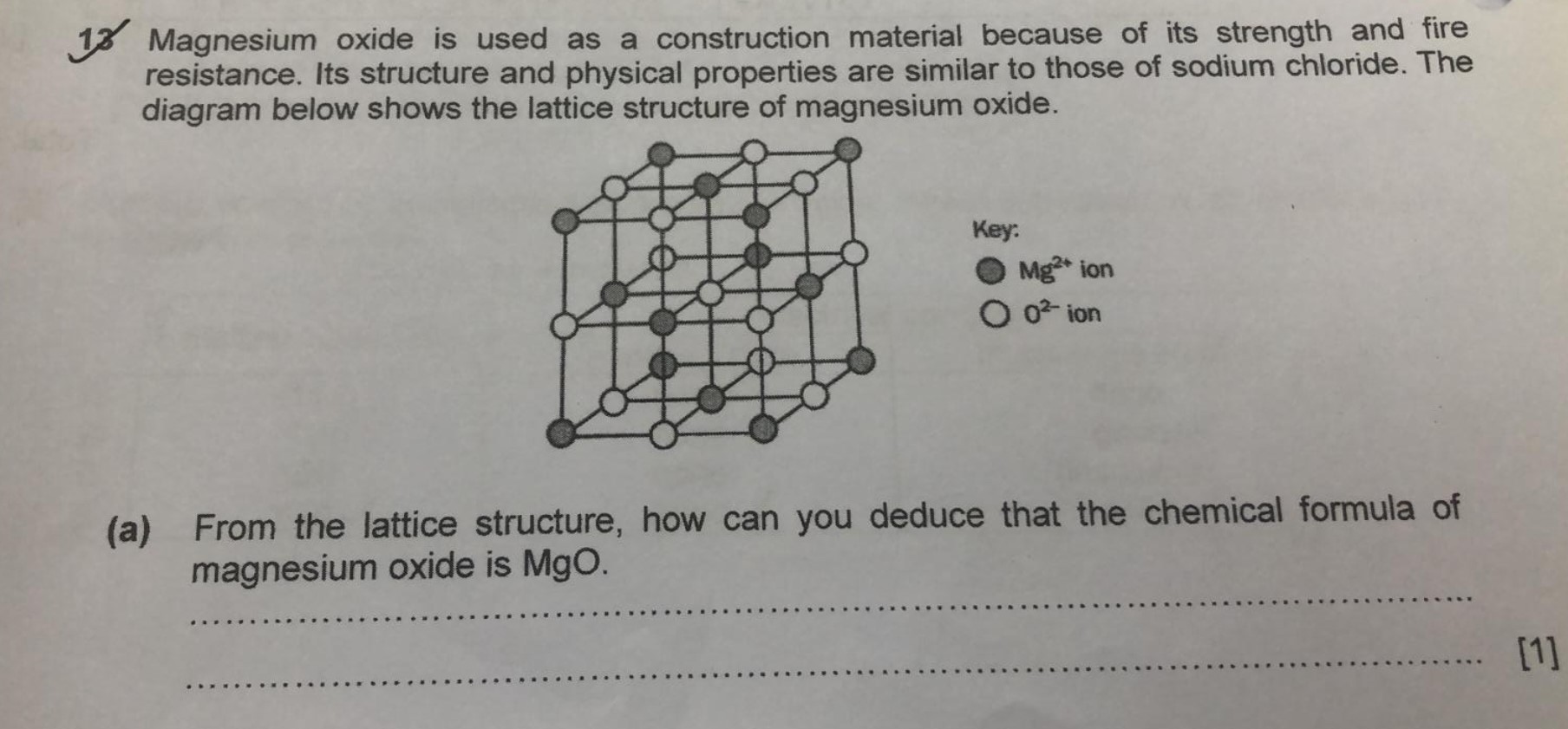
SOLVED: 13 Magnesium oxide is used as a construction material because of its strength and fire resistance. Its structure and physical properties are similar to those of sodium chloride. The diagram below
Magnesium Oxide: Percent Composition and Empirical Formula—ChemTopic™ Lab Activity | Flinn Scientific