![PDF] Asymmetric Introduction of Nucleophiles to the 2-Position of Pyrrolidine Ring through N-Acylpyrrolidinium Ion | Semantic Scholar PDF] Asymmetric Introduction of Nucleophiles to the 2-Position of Pyrrolidine Ring through N-Acylpyrrolidinium Ion | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/39b687777e953138690b0c5788cf843805df7214/3-Figure1-1.png)
PDF] Asymmetric Introduction of Nucleophiles to the 2-Position of Pyrrolidine Ring through N-Acylpyrrolidinium Ion | Semantic Scholar
Diastereoselective synthesis of functionalized pyrrolidines through N -bromosuccinimide-induced aziridine ring expansion cascade of cinnamylaziridine ... - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C4OB01384K
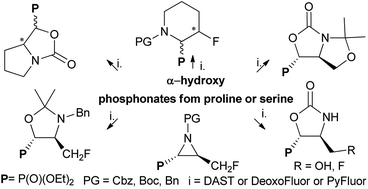
Pyrrolidine and oxazolidine ring transformations in proline and serine derivatives of α-hydroxyphosphonates induced by deoxyfluorinating reagents - RSC Advances (RSC Publishing)

EP0365324A1 - Spiro-isoquinoline-pyrrolidine tetrones and analogs thereof useful as aldose reductase inhibitors - Google Patents

Efficient Method for the Synthesis of Chiral Pyrrolidine Derivatives via Ring-Closing Enyne Metathesis Reaction | Organic Letters

1-(N-nitroamidino)pyrrolidine Structure - C5H10N4O2 - Over 100 million chemical compounds | Mol-Instincts

Locked Conformations for Proline Pyrrolidine Ring: Synthesis and Conformational Analysis of cis- and trans-4-tert-Butylprolines | The Journal of Organic Chemistry
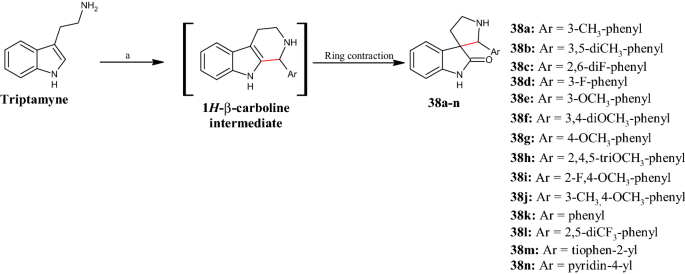
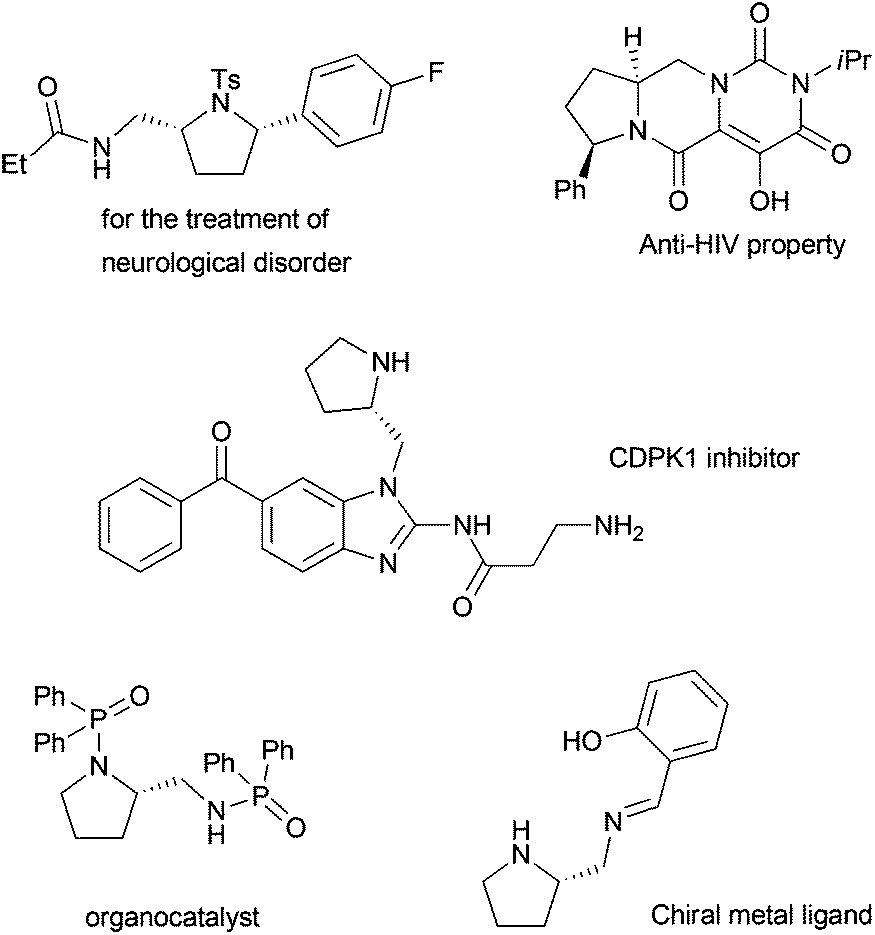
Pyrrolidine in Drug Discovery: A Versatile Scaffold for Novel Biologically Active Compounds | SpringerLink

a) Energy diagram of mechanism for formation of pyrrolidine ring from... | Download Scientific Diagram

3-Pyrroline, tech. 85% (remainder pyrrolidine), Thermo Scientific Chemicals, Quantity: 1 g | Fisher Scientific







![PDF] Use of Piperidine and Pyrrolidine in Knoevenagel Condensation | Semantic Scholar PDF] Use of Piperidine and Pyrrolidine in Knoevenagel Condensation | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/204d8cfb50b6267ae364d6e6a5a3077f018f4e9a/2-Figure2-1.png)