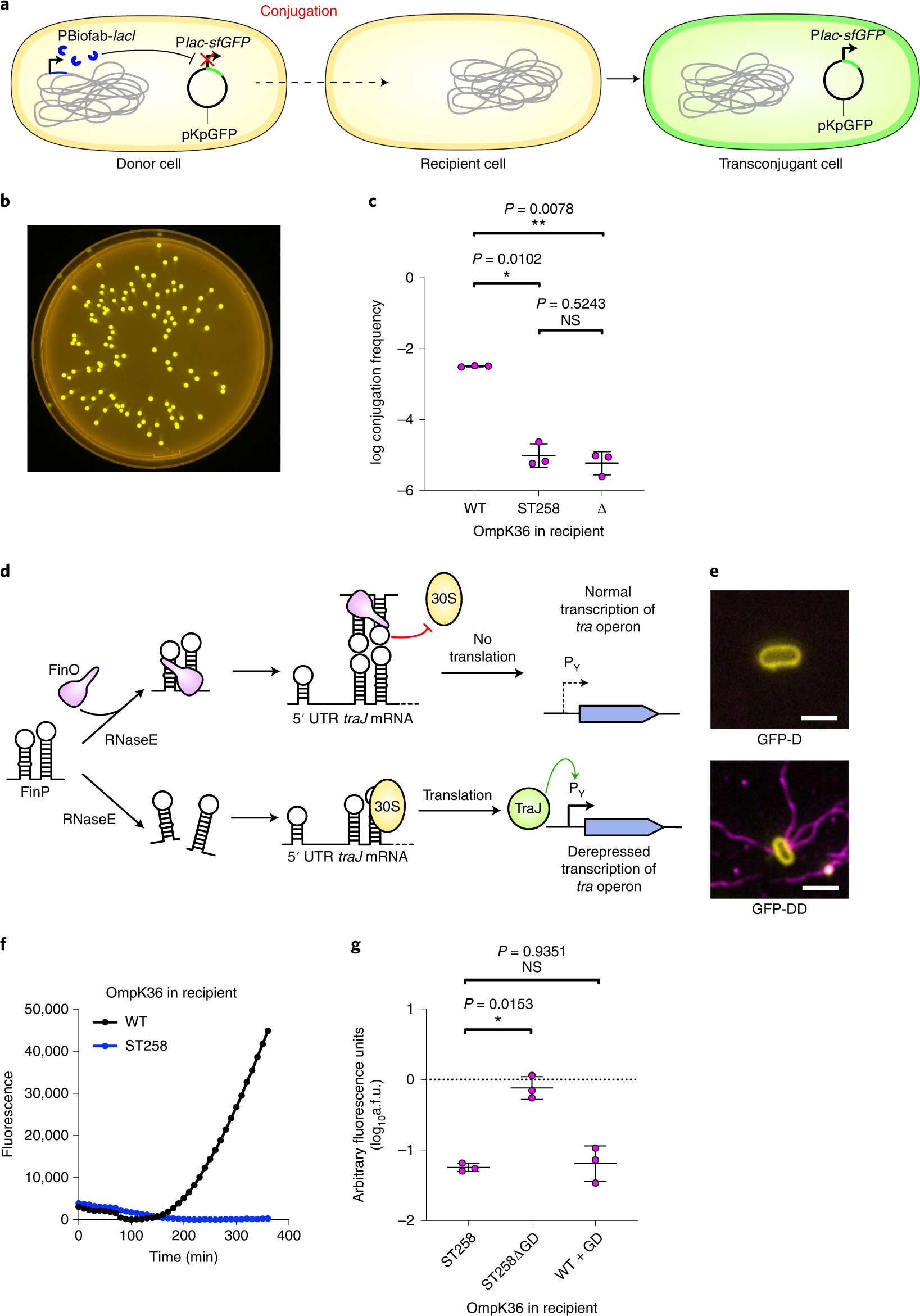
Novel two-chain structure utilizing KIRS2/DAP12 domain improves the safety and efficacy of CAR-T cells in adults with r/r B-ALL: Molecular Therapy - Oncolytics
![Non-Delivery Report: [550 5.4.1 Recipient address rejected: Access denied] – Exclaimer Cloud Knowledge Base Non-Delivery Report: [550 5.4.1 Recipient address rejected: Access denied] – Exclaimer Cloud Knowledge Base](https://exclaimerkbimages.blob.core.windows.net/images/Exclaimer%20Cloud/Signature%20Rules/NDR_MSActiveDirectory_OptiionalFeatures.png)
Non-Delivery Report: [550 5.4.1 Recipient address rejected: Access denied] – Exclaimer Cloud Knowledge Base

Safety and immunogenicity of INO-4800 DNA vaccine against SARS-CoV-2: A preliminary report of an open-label, Phase 1 clinical trial - eClinicalMedicine

A single dose of SARS-CoV-2 FINLAY-FR-1A vaccine enhances neutralization response in COVID-19 convalescents, with a very good safety profile: An open-label phase 1 clinical trial - The Lancet Regional Health – Americas
User Guide for AsyncOS 11.1 for Cisco Email Security Appliances - GD (General Deployment) - Understanding the Email Pipeline [Cisco Secure Email Gateway] - Cisco