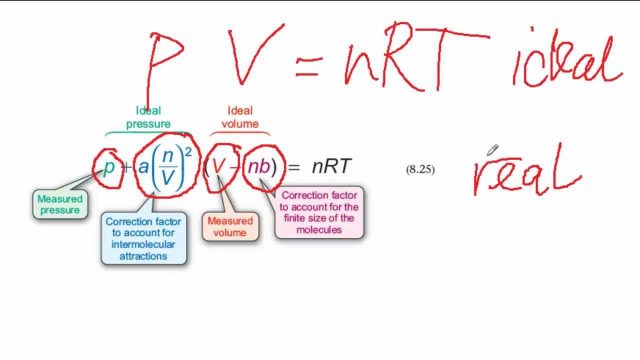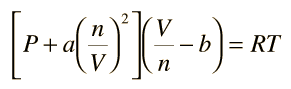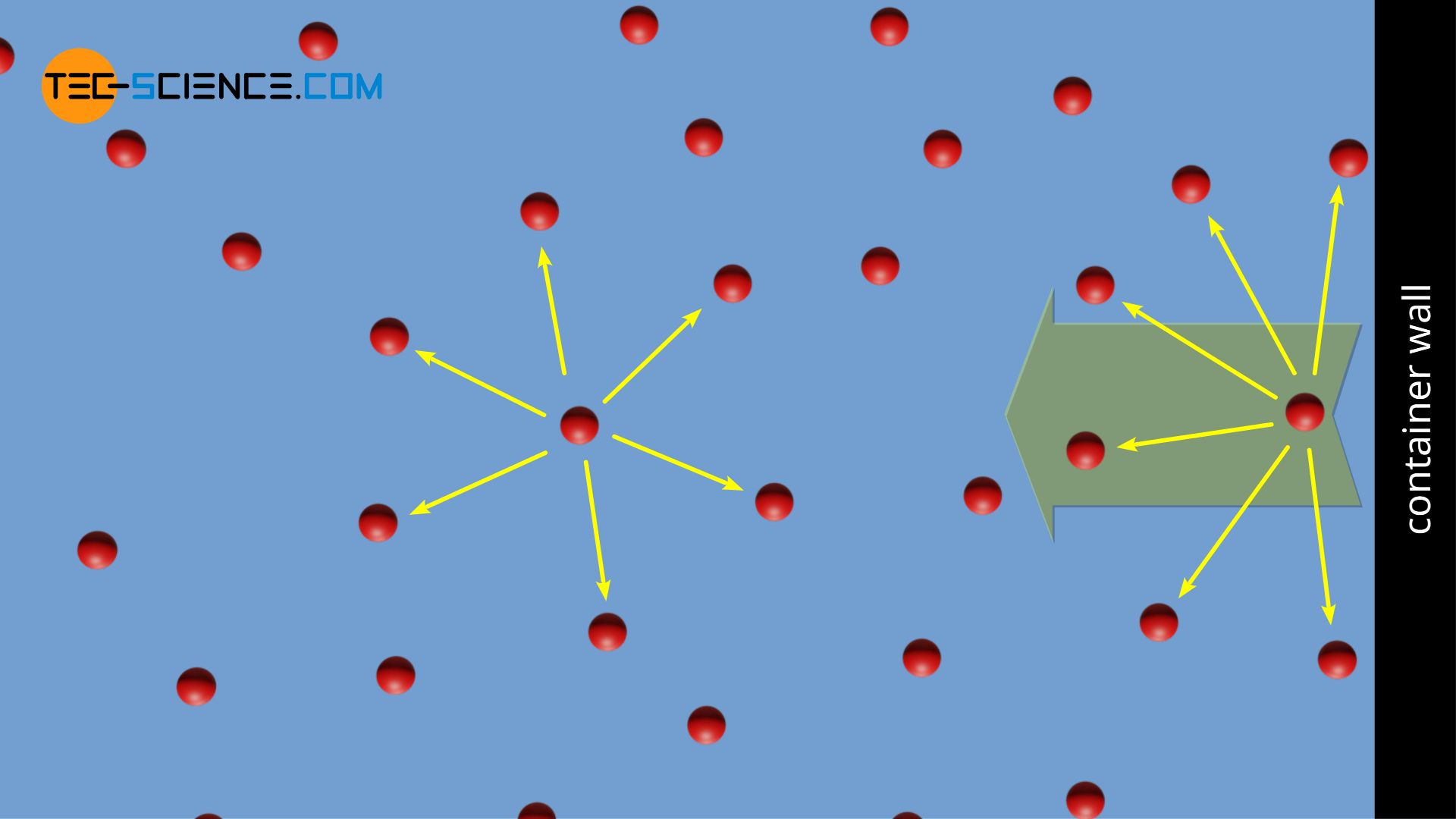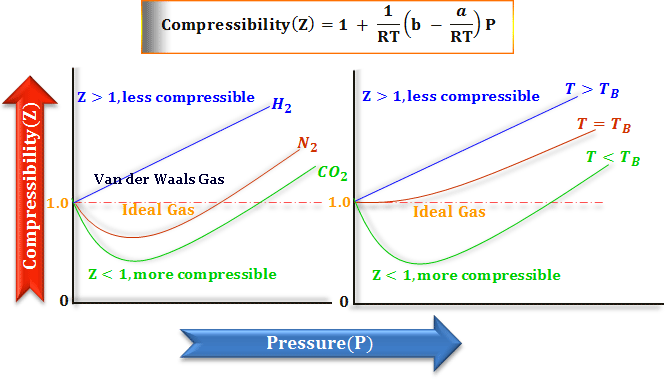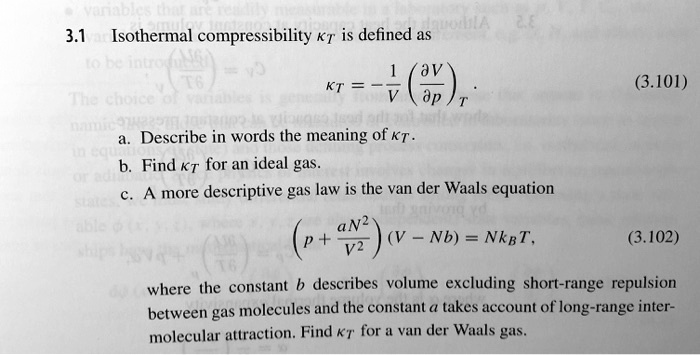
SOLVED: 3.1 Isothermal compressibility KT is defined a5 KT = (ov); (3.101) Describe in words the meaning of KT Find KT for an ideal gas more deseriptive gas law is the van

What is the major factor that accounts for most of the difference in these two values of pressure (ideal gas law vs. van der Waals equation)? | Socratic

Physics_in - Van der Waals equation of state ▫Share this post with friends.👍 . . . . . . #physics #fizik #fysik #fisica #физика #物理 #mathematics #science #ciencia #classes #chemistry #astronomy #space #