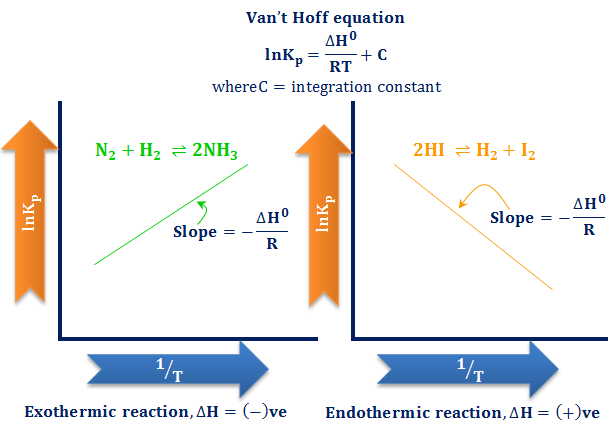
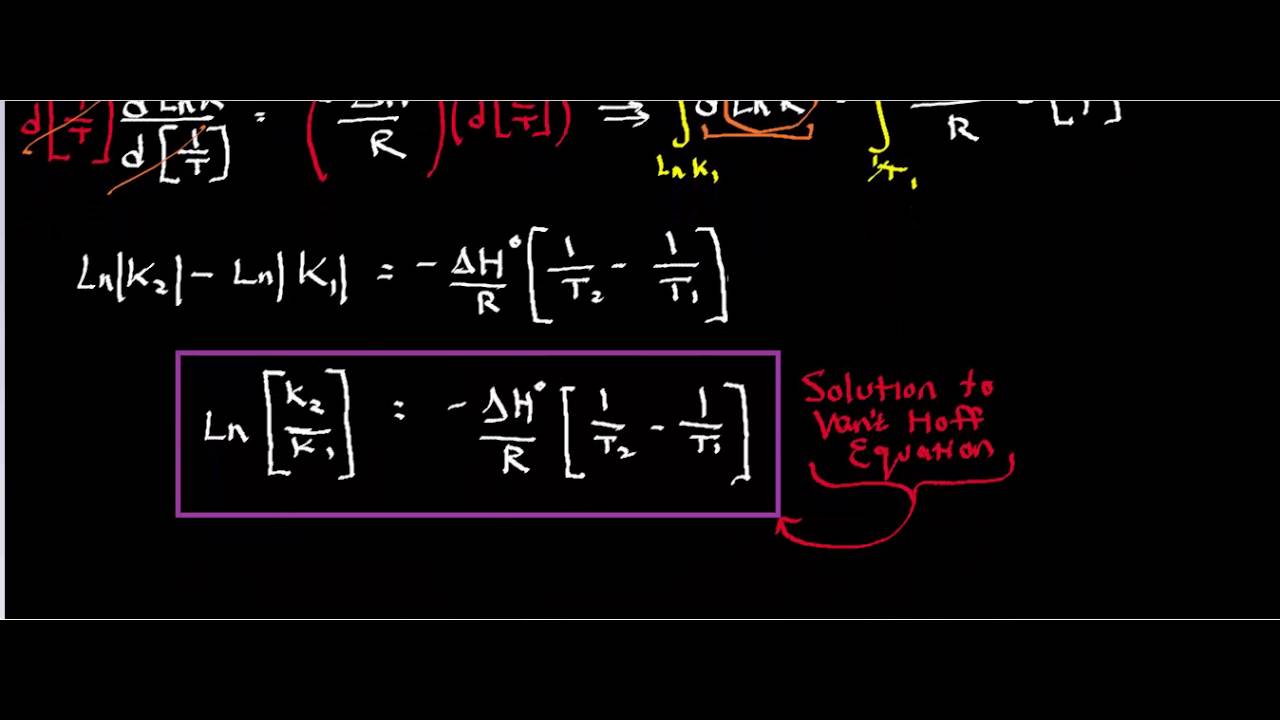
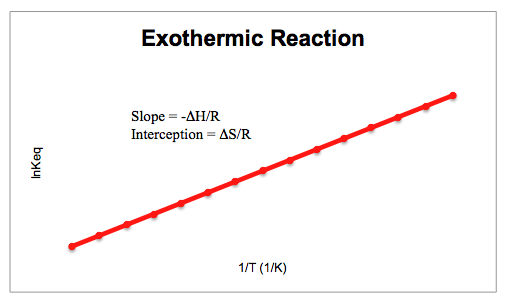
Jacobus Henricus van 't Hoff - Integrated van't Hoff equation (if the standard reaction enthalpy ΔH0 is known and constant in the temperature range of the measurement) is used for temperature correction.

SOLVED: a) The van't Hoff equation: A,Ho 4,So In K = RT relates the change in temperature to the chemical equilibrium constant Derive the van't Hoff equation starting with the expression for